| SECURITIES AND EXCHANGE COMMISSION | ||
| WASHINGTON, D.C. 20549 | ||
| FORM
|
| For
the fiscal year ended |
| OR |
| For the transition period from _____________ to _____________ |
| Commission
File No. |

| (Exact name of registrant as specified in its charter) |
| (State of incorporation) | (IRS Employer Identification Number) | |
| (Address of principal executive offices) | (Zip Code) |
|
|
||
| (Registrant's Telephone Number, including area code) |
Securities Registered pursuant to Section 12(b) of the Act
| Title of Each Class | Trading Symbol(s) | Exchange Registered | ||
Securities Registered pursuant to Section 12(g) of the Act
None
Indicate
by check mark if the registrant is a well-known seasoned issuer, as defined in Rule 405 of the Securities Act. Yes ☐
Indicate
by check mark if the registrant is not required to file reports pursuant to Section 13 or Section 15(d) of the Act. Yes ☐
| Page 1 |
Indicate
by check mark whether the registrant (1) has filed all reports required to be filed by Section 13 or 15(d) of the Securities Exchange
Act of 1934 during the preceding 12 months (or for such shorter period that the registrant was required to file such reports), and (2)
has been subject to such filing requirements for the past 90 days.
Indicate
by check mark whether the registrant (1) has submitted electronically and posted on its corporate website, if any, every Interactive
Data File required to be submitted and posted pursuant to Rule 405 of Regulation S-T (Section 232.405 of this chapter) during the preceding
12 months (or for such shorter period that the registrant was required to submit and post such files).
Indicate by check mark whether the registrant is a large accelerated filer, an accelerated filer, a non-accelerated filer, or a smaller reporting company. See definitions of “large accelerated filer”, “accelerated filer and “smaller reporting company” in Rule 12b-2 of the Exchange Act. (Check one):
| Large accelerated filer ☐ | Accelerated filer ☐ | |
| Smaller
reporting company |
Emerging
Growth Company |
Indicate
by check mark whether the registrant is a shell company (as defined in Rule 12b-2 of the Exchange Act). Yes ☐
The
aggregate market value of the shares of Common Stock held by non-affiliates as of December 31, 2022 based on the closing price of $16.75
per share on such date as reported on the NASDAQ System, was approximately $
As of September 8, 2023, shares of Common Stock, shares of Class B Common Stock, shares of Class C Common Stock and shares of Class A Non-voting Preferred Stock of the registrant were outstanding.
DOCUMENTS INCORPORATED BY REFERENCE
NONE
| Page 2 |
FONAR CORPORATION AND SUBSIDIARIES
TABLE OF CONTENTS
| Page 3 |
FONAR CORPORATION AND SUBSIDIARIES
PART I
ITEM 1. BUSINESS
GENERAL
FONAR Corporation, sometimes referred to as the “Company” or “FONAR”, is a Delaware corporation which was incorporated on July 17, 1978. Our address is 110 Marcus Drive, Melville, New York 11747 and our telephone number is 631-694-2929. FONAR also maintains a website at www.fonar.com. FONAR provides copies of its filings with the Securities and Exchange Commission on Forms 10-K, 10-Q and 8-K and amendments to these reports to stockholders on request.
We conduct our business in two segments. Our medical equipment segment is conducted directly through FONAR. Our physician management and diagnostic services segment is conducted through our subsidiary Health Management Corporation of America (“HMCA”). HMCA provides management services, administrative services, billing and collection services, credentialing services, contract negotiations, compliance consulting, purchasing, IT services, hiring, conducting interviews and managing personnel, storage of medical records, office space, equipment, repair, maintenance service, and clerical and other non-medical personnel to medical providers engaged in diagnostic imaging. In addition to acting as a management company, HMCA owns and operates five diagnostic imaging facilities in Florida, where the corporate practice of medicine is permitted.
FONAR is engaged in the business of designing, manufacturing, selling and servicing magnetic resonance imaging scanners, also referred to as “MRI” or “MR” scanners, which utilize MRI technology for the detection and diagnosis of human disease, abnormalities, other medical conditions and injuries. FONAR’s founders built the first MRI scanner in 1977 and FONAR introduced the first commercial MRI scanner in 1980. FONAR is also the originator of the iron-core non-superconductive and permanent magnet MRI technology.
FONAR’s iron frame technology made FONAR the originator of “open” MRI scanners. We introduced the first “open” MRI in 1980. Since that time we have concentrated on further application of our “open” MRI, introducing most recently the Upright® Multi-Position™” MRI scanner (also referred to as the “Upright®” or “Stand-Up®” MRI scanner) and the FONAR 360™ MRI scanner. The FONAR 360™ MRI is not presently being marketed.
See Note 16 to the Consolidated Financial Statements for separate financial information regarding our medical equipment and physician and diagnostic management services segments.
| Page 4 |
FONAR CORPORATION AND SUBSIDIARIES
CAUTIONARY NOTE REGARDING FORWARD LOOKING STATEMENTS.
Certain statements made in this Annual Report on Form 10-K are “forward-looking statements”, within the meaning of the Private Securities Litigation Reform Act of 1995, regarding the plans and objectives of Management for future operations. Such statements involve known and unknown risks, uncertainties and other factors that may cause our actual results, performance or achievements to be materially different from any future results, performance or achievements expressed or implied by such forward-looking statements. These forward-looking statements are based on current expectations that involve numerous risks and uncertainties. Our plans and objectives are based, in part, on assumptions involving the expansion of business. These assumptions involve judgments with respect to, among other things, future economic, competitive and market conditions and future business decisions, all of which are difficult or impossible to predict accurately and many of which are beyond our control. Although we believe that our assumptions underlying the forward-looking statements are reasonable, any of the assumptions could prove inaccurate and, therefore, there can be no assurance that the forward-looking statements included in this Annual Report will prove to be accurate. In light of the significant uncertainties inherent in our forward-looking statements, the inclusion of such information should not be regarded as a representation by us or any other person that our objectives and plans will be achieved.
MEDICAL EQUIPMENT SEGMENT
PRODUCTS
The Upright® MRI scanner is the product we are presently promoting.
The Upright® MRI is a “whole-body” MRI, meaning that it can be used to scan virtually any part of the body.
The Upright® MRI differs from conventional MRI scanners in that it is not limited to scanning patients in the recumbent posture. For example, patients can be scanned while sitting, standing, bending, or lying down.
The Upright® MRI is also, by design, a non-claustrophobic MRI scanner. The Upright® MRI employs a dipole magnet whose magnetic field orientation is transverse to the axis of the patient’s body. The gap between the poles of the magnet is the space into which the patient is placed. Because the magnetic field direction is horizontal and transverse to the body, a patient who is scanned seated or standing has an unobstructed view out of the gap of the magnet. In typical installations, patients watch television while being scanned, without the aid of special glasses with mirrors.
The fact that the patient space is unobstructed permits scanning in a variety of postures that cannot be duplicated in conventional MRI scanners. Most conventional MRI scanners in use today employ solenoidal super-conducting magnets whose magnetic field orientation is along the axis of the patient’s body, which must be placed into the bore of the scanner in either a supine or prone posture. Our experience is that when presented with a choice between being scanned lying down in a tunnel-like enclosure or seated in an open MRI, most patients will choose the latter.
| Page 5 |
FONAR CORPORATION AND SUBSIDIARIES
The Upright® MRI facilitates patient scanning in a variety of postures thanks to a unique, three-axis patient handling system. The motorized patient table, or bed, can be rotated to any angle between 0 (horizontal) and 84 degrees (nearly vertical). Unlike a conventional recumbent MRI patient table, which can only move into or out of the scanner’s bore, the Upright® MRI bed can be translated with two degrees of freedom, in/out and up/down. User-friendly software allows the scanner operator to move the anatomical region of interest precisely to the center of the magnet using a cursor placed on a localizer image. Anatomically true image orientation is assured, regardless of the rotation angle of the bed, via computer read-back of the table’s position. A seat can be hooked onto the bed in a variety of locations, or removed, as needed. Transpolar VersaRests™ and other devices can be used to keep the patient comfortable and motionless throughout the scanning process.
IMAGE QUALITY AND FIELD STRENGTH
Most commercially available MRI scanners range in magnetic field strength from about 0.2 T (Tesla) to 7.0 T, and open MRI scanners range from about 0.2 T to 1.2 T.
Field strength is an important characteristic of MRI scanners, but not the only one. Higher field strengths generally provide higher signal-to-noise ratios (SNR) on account of the Boltzmann distribution, but SNR is not the only determinant of image quality. For example, the spin-lattice relaxation time T1 that characterizes the nuclear magnetic resonance (NMR) signal increases with field strength, decreasing the difference in T1 values between tissues that is an essential contributor to contrast in images. For example, grey/white matter contrast in the brain falls off rapidly above about 1.0 T, and some studies have shown that optimal tissue contrast occurs in the mid-field region, down to 0.2 T. Imaging bandwidth, receiver coil design, pulse sequence design, and scan parameters significantly affect image quality. Indeed, researchers and MRI vendors are pushing the boundaries of MRI technology in both directions, that is, to very low (1 – 199 mT) and very high (7.0 T and above) field strengths for a variety of technical and diagnostic reasons. For instance, one advantage of lower field strengths is that image artifacts arising from metallic implants such as surgical screws diminish as field strength decreases. This is particularly important for surgeons referring their postoperative patients for diagnostic imaging studies.
The Upright® MRI operates at a mid-field strength of 0.6 T and enjoys wide acceptance in the radiological community. The scanner is diagnostically versatile and equipped with a broad range of clinically proven imaging protocols that produce images of exceptional quality, and a fully-featured, robust, and user-friendly software interface.
DIAGNOSTIC ADVANTAGES OF POSITIONAL MRI
Apart from its attractiveness as an open, non-claustrophobic, general-purpose MRI, the Upright® MRI can deliver diagnostically relevant information that correlates with patient posture.
For example, a variety of injuries to and pathologies of the spine, such as spondylolisthesis (“slipped disc”), may go undetected in the recumbent posture, but manifest themselves when the patient is scanned in a normal, weight-bearing (“physiological”) position, such as seated, or seated in forward flexion, extension, or standing.
| Page 6 |
FONAR CORPORATION AND SUBSIDIARIES
The Upright® MRI has demonstrated its value for patients suffering from scoliosis, who typically undergo regular x-ray exams over a course of years. A study by the National Cancer Institute (2000) of 5,466 women with scoliosis reported a 70% increase in breast cancer resulting from 24.7 chest x-rays received on average over the course of their treatment. Prior to the advent of the Upright® MRI, the x-ray machine was the only imaging modality that could evaluate the condition because the patient must be imaged standing. FONAR has developed an RF receiver coil and a 3D scanning protocol that for the first time allows scoliosis patients to obtain diagnostic, multi-slice images of their spines while standing, without the risks associated with radiation, and with the soft-tissue-contrast benefits of MRI over x-ray.
The utility of upright, weight-bearing MRI is not limited to the spine. For example, approximately one in a thousand people (some 200,000 to 500,000 in the US) have a congenital condition known as Chiari malformation, an abnormality of the brain at the junction with the spine at the base of the skull. In people with Chiari malformation, the lowest lying structures of the brain, the tonsils of the cerebellum, descend into and become entrapped by the foramen magnum, the circular bony opening at the base of the skull where the spinal cord exits. While most of these individuals are asymptomatic, many suffer from more severe forms of the syndrome (e.g., type II or Arnold-Chiari syndrome), in which brain stem compression results in severe neurological symptoms. The Chiari syndrome is also called Cerebellar Tonsillar Ectopia (CTE) because of the displacement (ectopia) of the cerebellar tonsils. Classic symptoms of Chiari syndrome include the “drop attack,” in which the afflicted individual unexpectedly experiences an explosive rush at the base of the brain that runs down the body to the extremities, causing the patient to collapse in a temporary neuromuscular paralysis. These symptoms subside when the patient is lying down. Conventional lie-down MRI scanners cannot make an adequate evaluation of the pathology since this pathology is most visible and the symptoms are most acute when the patient is scanned in the upright, weight-bearing position (Brain Injury 2010, 24 (7-8) 988-994).
In the body, the Upright® MRI is being utilized in a variety of ways, for example to image pelvic organ prolapse in the standing posture, inguinal hernias, defecation in the sitting posture (utilizing cine MRI), and the prostate in the sitting posture (utilizing a flat, multi-channel receiver coil on top of which the patient simply sits).
PRODUCT MARKETING
FONAR’s principal marketing efforts in the medical equipment segment have been focused on the Upright® MRI, which we believe is a unique product. We expect to focus on the Upright® MRI going forward.
The principal markets for the Company’s scanners are private diagnostic imaging centers and hospital outpatient imaging facilities.
We use internal personnel and independent manufacturer’s representatives for domestic and foreign sales.
FONAR’s marketing strategy has been designed to reach key purchasing decision makers with information concerning the Upright® MRI. This has led to many inquiries and some sales of the Upright® MRI scanner and is intended to increase FONAR’s presence in the medical equipment market. FONAR focuses primarily on four target audiences: neurosurgeons, orthopedic surgeons, radiologists, and general physicians.
| Page 7 |
FONAR CORPORATION AND SUBSIDIARIES
Our advertising for FONAR and HMCA reinforces the unique value provided by the FONAR Upright® MRI scanner. We have increased internet awareness of our product by driving patient traffic to the HMCA scanning centers we manage via the FONAR website as well as through websites for each HMCA location. These websites give prospective customers of Upright® MRI scanners a view of operating Upright® MRI centers and highlight the benefits of using the Upright® MRI scanner. A complete list of the sites managed by HMCA can be found at HMCA’s website, www.hmca.com.
SERVICE AND UPGRADES FOR MRI SCANNERS
Income is generated from the installed base in two principal areas, namely, service and upgrades. Service and maintenance revenues from our external installed base were approximately $7.5 million in fiscal 2023 and $7.7 million in fiscal 2022.
We expect to maintain service revenues at present levels or better, based on the demonstrated longevity of the Upright® MRI scanner and continued customer satisfaction with the product. Critical to this longevity and customer satisfaction is the stream of software improvements and hardware upgrades that FONAR has delivered over the years to keep the scanners competitive with the latest technology in the marketplace. We also anticipate that our installed base of scanners will generate income from upgrades in future fiscal years.
RESEARCH AND DEVELOPMENT
During the fiscal year ended June 30, 2023, we incurred expenditures of $1,567,749, none of which were capitalized, on research and development, as compared to $1,494,181, none of which were capitalized, during the fiscal year ended June 30, 2022.
Research and development activities have focused principally on software improvements to the user interface of the MRI scanner. The Windows-based Sympulse™ platform controls all of the functions of the Upright® scanner except those of the versatile, multi-position patient table. Separate, dedicated, motion-control software is used to maneuver the Upright® bed, and development of this software is ongoing as well.
While software improvements to the user interface are important in their own right, significant value is added to the MRI scanner by the modification of existing protocols for examining various parts of the body, and the development of new protocols that utilize new underlying capabilities of the pulse sequence software. Over time, FONAR users have become accustomed to the steady improvement in the recommended clinical protocols that accompany new software releases. More significantly, in recent years we have seen increasing adoption of FONAR-recommended clinical protocols over those developed on site. This is a testament to the superior image quality they produce in attractively short scan times.
| Page 8 |
FONAR CORPORATION AND SUBSIDIARIES
The development of clinically practical scan protocols and software depends on close contact between research and development scientists and engineers, and end users. That close contact is facilitated in part by the relationship with HMCA and the scanning centers. In addition to that collaboration, R&D staff have pursued a variety of novel and Upright® MRI-specific research projects. It is anticipated that these will ultimately lead to new applications that are made available to existing customers as upgrade add-ons to their machines. For example, phase-contrast imaging techniques originally developed for angiography have recently been applied to cerebro-spinal fluid (CSF) flow. Analysis of CSF flow in upright and recumbent postures may prove to be of significant value in the evaluation of a variety of disorders and lead to a better understanding of human physiology.
BACKLOG
Our backlog of unfilled orders at September 8, 2023 was approximately $608,000, as compared to $844,000 at September 8, 2022. It is expected that the existing backlog of orders will be filled during the 2023 fiscal year.
PATENTS AND LICENSES
We currently have numerous patents in effect which relate to the technology and components of our MRI scanners. We believe that these patents, and the know-how we have developed, are material to our business.
One of our patents, issued in the name of Dr. Damadian and licensed to FONAR, was United States patent No. 3,789,832, Apparatus and Method for Detecting Cancer in Tissue, also referred to in this report as the “1974 Patent”. The 1974 Patent was the first MRI patent issued by the United States Patent Office. The development of our MRI scanners has been based upon the 1974 Patent, and we believe that the 1974 Patent was the first of its kind to utilize MR to scan the human body and to detect cancer. The 1974 Patent was extended beyond its original 17-year term and expired in February, 1992. A number of FONAR’s existing patents specifically relate to protecting FONAR’s position in the Upright MRI market. The patents further enhance Dr. Damadian’s pioneer patent, the 1974 Patent, that initiated the MRI industry and provided the original invention of MRI scanning. The terms of the patents in FONAR’s portfolio extend to various times.
We have significantly enhanced our patent position within the industry and now possess a substantial patent portfolio which provides us, under the aegis of United States patent law, “the exclusive right to make, use and sell” many of the scanner features which FONAR pioneered and which are now incorporated in most MRI scanners sold by the industry. As of June 30, 2023, 225 patents had been issued to FONAR, and approximately 8 patents were pending. Two new patents were issued in fiscal year 2023. One patent describes an equipment calibration system for ultrasound equipment used to non-invasively measure intracranial pressure. The other described a method for identifying the presence and amount of vascular congestion using MRI.
We also have patent cross-licensing agreements with other MRI manufacturers. We have not licensed, however, any technology relating to Upright® MRI scanning.
| Page 9 |
FONAR CORPORATION AND SUBSIDIARIES
PRODUCT COMPETITION
MRI SCANNERS
FONAR faces competition for MRI product sales from companies such as Siemens, General Electric, Hitachi, Philips, Canon, and United Imaging. Each of these is primarily focused on the high-field (1.0 T and above) marketplace, though some have produced open MRI scanners for imaging in the recumbent posture. None of these firms has so far introduced an open, upright MRI.
In recent years Paramed and Esaote have introduced MRI scanners aimed at the upright, weight-bearing MRI market. Their success in the US has so far been limited. We believe that the higher field strength and larger dimensions of the FONAR Upright® MRI magnet, together with the greater variety of patient positioning possibilities afforded by the FONAR Upright® MRI bed, give us a competitive advantage over the products introduced by these companies.
Most of our competitors have marketing and financial resources more substantial than those available to us. They have in the past, and may in the future, heavily discount the sales price of their scanners.
OTHER IMAGING MODALITIES
FONAR’s MRI scanners also compete with other diagnostic imaging systems, all of which are based upon the ability of some form of energetic wave to penetrate human tissue and be detected by either photographic film or electronic devices for presentation on a display monitor. Three different kinds of energy waves – x-ray, gamma, and sound – are used in medical imaging techniques that compete with MRI, the first two of which involve exposing the patient to potentially harmful radiation. These other imaging modalities compete with MRI products on the basis of cost, space requirements, and specific clinical applications.
X-rays are the most common energy source used in imaging the body and are employed in three imaging modalities: conventional x-ray systems, computerized tomography (CT), and digital radiography. None of these enjoy the exquisite soft-tissue contrast of MRI, but they do offer high resolution imaging in certain applications and high speed of image acquisition.
Nuclear medicine systems, which are based upon the detection of photons (gamma radiation) generated by radioactive pharmaceuticals introduced into the body, are used to provide information concerning soft tissue and internal body organs and particularly to examine organ function over time.
Ultrasound systems emit, detect, and process high frequency sound waves reflected from organ boundaries and tissue interfaces to generate images of soft tissue and internal body organs. Although the images are substantially less detailed than those obtainable with x-rays or MRI, ultrasound is generally considered harmless and therefore has found applications in imaging the pregnant uterus and the breast, to name two.
| Page 10 |
FONAR CORPORATION AND SUBSIDIARIES
X-ray (including CT), nuclear medicine, and ultrasound compete with the MRI scanners by offering significantly lower price and space requirements. However, history has shown that the superior tissue contrast characteristics of MRI have secured its place as the diagnostic imaging modality of choice for a wide variety of pathologies.
GOVERNMENT REGULATION
FDA Regulation
The Food and Drug Administration in accordance with Title 21 of the Code of Federal Regulations regulates the manufacturing and marketing of FONAR’s MRI scanners. The regulations can be classified as either pre-market or post-market. The pre-market requirements include obtaining marketing clearance, proper device labeling, establishment registration and device listing. Once the products are on the market, FONAR must comply with post-market surveillance controls. These requirements include the Quality Systems Regulation, or “QSR”, also known as Current Good Manufacturing Practices or CGMPs, and Medical Device Reporting, also referred to as MDR regulations. The QSR is a quality assurance requirement that covers the design, packaging, labeling and manufacturing of a medical device. The MDR regulation is an adverse event-reporting program.
Classes of Products
Under the Medical Device Amendments of 1976 to the Federal Food, Drug and Cosmetic Act, all medical devices are classified by the FDA into one of three classes. A Class I device is subject only to general controls, such as labeling requirements and manufacturing practices; a Class II device must comply with certain performance standards established by the FDA; and a Class III device must obtain pre-market approval from the FDA prior to commercial marketing. FONAR’s products are Class II devices. Class II devices are subject to “General Controls”; General Controls include:
| 1. | Establishment registration of companies which are required to register under 21 CFR Part 807.20, such as manufacturers, distributors, re-packagers and re-labelers. |
| 2. | Medical device listing with FDA of devices to be marketed. |
| 3. | Manufacturing devices in accordance with the Current Good Manufacturing Practices Quality System Regulation in 21 CFR Part 820. |
| 4. | Labeling devices in accordance with labeling regulations in 21 CFR Part 801 or 809. |
| 5. | Submission of a Premarket Notification, pursuant to 510(k), before marketing a device. |
In addition to complying with general controls, Class II devices are also subject to special controls. Special controls may include special labeling requirements, guidance documents, mandatory performance standards and post-market surveillance.
On October 3, 2000 FONAR received FDA clearance for the Upright® MRI under the name “Indomitable”.
| Page 11 |
FONAR CORPORATION AND SUBSIDIARIES
Premarketing Submission
Each person who wants to market Class I, II and some III devices intended for human use in the U.S. must submit a 510(k) to FDA at least 90 days before marketing unless the device is exempt from 510(k) requirements. A 510(k) is a pre-marketing submission made to FDA to demonstrate that the device to be marketed is as safe and effective, that is, substantially equivalent, SE, to a legally marketed device that is not subject to pre-market approval, PMA. Applicants must compare their 510(k) device to one or more similar devices currently on the U.S. market and make and support their substantial equivalency claims.
The FDA is committed to a 90-day clearance after submission of a 510(k), provided the 510(k) is complete and there is no need to submit additional information or data.
The 510(k) is essentially a brief statement and description of the product. As FONAR’s scanner products are Class II products, there are no pre-market data requirements.
An investigational device exemption, also referred to as IDE, allows the investigational device to be used in a clinical study pending FDA clearance in order to collect safety and effectiveness data required to support the Premarket Approval, also referred to as PMA, application or a Premarket Notification pursuant to 510(k), submission to the FDA. Clinical studies are most often conducted to support a PMA.
For the most part, however, we have not found it necessary to utilize IDE’s. The standard 90 day clearance for our new MRI scanner products classified as Class II products makes the IDE unnecessary, particularly in view of the time and effort involved in compiling the information necessary to support an IDE.
Quality System Regulation
The Quality Management System is applicable to the design, manufacture, administration of installation and servicing of magnetic resonance imaging scanner systems. The FDA has authority to conduct detailed inspections of manufacturing plants, to establish Good Manufacturing Practices which must be followed in the manufacture of medical devices, to require periodic reporting of product defects and to prohibit the exportation of medical devices that do not comply with the law.
Medical Device Reporting Regulation
Manufacturers must report all MDR reportable events to the FDA. Each manufacturer must review and evaluate all complaints to determine whether the complaint represents an event which is required to be reported to FDA. Section 820.3(b) of the Quality Systems regulation defines a complaint as, “any written, electronic or oral communication that alleges deficiencies related to the identity, quality, durability, reliability, safety, effectiveness, or performance of a device after it is released for distribution.”
| Page 12 |
FONAR CORPORATION AND SUBSIDIARIES
A report is required when a manufacturer becomes aware of information that reasonably suggests that one of their marketed devices has or may have caused or contributed to a death, serious injury, or has malfunctioned and that the device or a similar device marketed by the manufacturer would be likely to cause or contribute to a death or serious injury if the malfunction were to recur.
Malfunctions are not reportable if they are not likely to result in a death, serious injury or other significant adverse event experience.
A malfunction which is or can be corrected during routine service or device maintenance still must be reported if the recurrence of the malfunction is likely to cause or contribute to a death or serious injury if it were to recur.
We have established and maintained written procedures for implementation of the MDR regulation. These procedures include internal systems that:
provide for timely and effective identification, communication and evaluation of adverse
events;
provide a standardized review process and procedures for determining whether or not an
event is reportable; and
provide procedures to insure the timely transmission of complete reports.
These procedures also include documentation and record keeping requirements for information that was evaluated to determine if an event was reportable;
all medical device reports and information submitted to the FDA;
any information that was evaluated during preparation of annual certification reports; and
systems that ensure access to information that facilitates timely follow up and inspection by
FDA.
FDA Enforcement
FDA may take the following actions to enforce the MDR regulation:
FDA-Initiated or Voluntary Recalls
Recalls are regulatory actions that remove a hazardous, potentially hazardous, or a misbranded product from the marketplace. Recalls are also used to convey additional information to the user concerning the safe use of the product. Either FDA or the manufacturer can initiate recalls.
There are three classifications, i.e., I, II, or III, assigned by the Food and Drug Administration to a particular product recall to indicate the relative degree of health hazard presented by the product being recalled.
| Page 13 |
FONAR CORPORATION AND SUBSIDIARIES
Class I
Is a situation in which there is a reasonable probability that the use of, or exposure to, a violative product will cause serious adverse health consequences or death.
Class II
Is a situation in which use of, or exposure to, a violative product may cause temporary or medically reversible adverse health consequences or where the probability of serious adverse health consequences is remote.
Class III
Is a situation in which use of, or exposure to, a violative product is not likely to cause adverse health consequences.
FONAR has initiated six voluntary recalls, which occurred between 1987-2016. Five of the recalls were Class II and one was Class III. The recalls involved making minor corrections to the product in the field. Frequently, corrections which are made at the site of the device are called field corrections as opposed to recalls.
Civil Money Penalties
The FDA, after an appropriate hearing, may impose civil money penalties for violations of the FD&C Act that relate to medical devices. In determining the amount of a civil penalty, FDA will take into account the nature, circumstances, extent, and gravity of the violations, the violator’s ability to pay, the effect on the violator’s ability to continue to do business, and any history of prior violations.
Warning Letters
FDA issues written communications to a firm, indicating that the firm may incur more severe sanctions if the violations described in the letter are not corrected. Warning letters are issued to cause prompt correction of violations that pose a hazard to health or that involve economic deception. The FDA generally issues the letters before pursuing more severe sanctions.
Seizure
A seizure is a civil court action against a specific quantity of goods which enables the FDA to remove these goods from commercial channels. After seizure, no one may tamper with the goods except by permission of the court. The court usually gives the owner or claimant of the seized merchandise approximately 30 days to decide a course of action. If they take no action, the court will recommend disposal of the goods. If the owner decides to contest the government’s charges, the court will schedule the case for trial. A third option allows the owner of the goods to request permission of the court to bring the goods into compliance with the law. The owner of the goods is required to provide a bond or, security deposit, to assure that they will perform the orders of the court, and the owner must pay for FDA supervision of any activities by the company to bring the goods into compliance.
| Page 14 |
FONAR CORPORATION AND SUBSIDIARIES
Citation
A citation is a formal warning to a firm of intent to prosecute the firm if violations of the FD&C Act are not corrected. It provides the firm an opportunity to convince FDA not to prosecute.
Injunction
An injunction is a civil action filed by FDA against an individual or company. Usually, FDA files an injunction to stop a company from continuing to manufacture, package or distribute products that are in violation of the law.
Prosecution
Prosecution is a criminal action filed by FDA against a company or individual charging violation of the law for past practices.
Foreign and Export Regulation
We obtain approvals as necessary in connection with the sales of our products in foreign countries. In some cases, FDA approval has been sufficient for foreign sales as well. Our standard practice has been to require either the distributor or the customer to obtain any such foreign approvals or licenses which may be required.
Legally marketed devices that comply with the requirements of the Food Drug & Cosmetic Act require a Certificate to Foreign Government issued by the FDA for export. Other devices that do not meet the requirements of the FD&C Act but comply with the laws of a foreign government require a Certificate of Exportability issued by the FDA. All products which we sell have FDA clearance and would fall into the first category.
Foreign governments have differing requirements concerning the import of medical devices into their respective jurisdictions. The European Union’s new medical device regulation, EU 2017/745 went into effect on May 25, 2021, and contains significant changes from the prior European regulatory scheme. We have applied to the Notified Body, TUV-SUD, to perform a Conformity Assessment of our technical documentation and our Quality Management System. We (optimistically) expect to have this completed by end of our third quarter, March 30, 2024.
Other countries require that their own testing laboratories perform an evaluation of our devices. This requires that we must bring the foreign agency’s personnel to the USA to perform the evaluation at our expense before exporting.
Some countries, including many in Latin America and Africa, have very few regulatory requirements, beyond FDA clearance.
To date, FONAR has been able to comply with all foreign regulatory requirements applicable to its export sales.
| Page 15 |
FONAR CORPORATION AND SUBSIDIARIES
PHYSICIAN AND DIAGNOSTIC SERVICES MANAGEMENT BUSINESS
Health Diagnostics Management, LLC (HDM) is owned by Health Management Corporation of America (70.8%) and investors (29.2%). Health Management Corporation of America is owned 100% by FONAR Corporation. During the fiscal year 2022, the Company purchased non-controlling interests from the minority shareholders for $546,000.
HDM operates under the assumed name “Health Management Company of America” (“HMCA”).
The combined business (HDM and Health Management Corporation of America) will be referred to as “HMCA” for all periods before and after July 1, 2015, unless otherwise indicated.
HMCA provides comprehensive non-medical management services to diagnostic imaging facilities. These services include administrative services, billing and collection services, credentialing services, contract negotiations, compliance consulting, purchasing IT services, hiring, conducting interviews, training, supervision and management of non-medical personnel, storage of medical records, office space, equipment, repair maintenance services, accounting, assistance with compliance matters and the development and implementation of practice growth and marketing strategies.
As of June 30, 2023, HMCA managed a total of 41 MRI scanners of which twenty-four (24) scanners are located in New York and seventeen (17) scanners are located in Florida. For the 2023 fiscal year, the revenues HMCA recognized from the MRI facilities has increased to $90.4 million from $89.4 million in fiscal 2022. Six of the facilities in Florida are owned by HMCA subsidiaries, where the corporate practice of medicine is permitted.
We believe the utilization of FONAR Upright® MRI scanning systems, which are produced under the protection of our patents, accounts for the historically robust patient volume at the scanning facilities. During fiscal 2023, two scanners were installed in Casselberry, Florida. We completed the consolidation of our two Manhattan centers into their new location in Midtown, removing the scanner previously located at Avenue A. The extremity-only scanner at our Brooklyn location was deemed to be passed its useful life and was removed from service.
HMCA GROWTH STRATEGY
HMCA’s growth strategy focuses on upgrading and expanding the existing facilities it manages and expanding the number of facilities it either owns or manages for its clients, including new sites. In connection with improving the performance of the facilities, we have added high field MRI scanners, extremity scanners and x-ray machines to the Upright® MRI scanners at certain of the sites where such additional diagnostic imaging modalities are expected to produce the greatest return.
| Page 16 |
FONAR CORPORATION AND SUBSIDIARIES
PHYSICIAN AND DIAGNOSTIC MANAGEMENT SERVICES
HMCA’s services to the facilities it manages encompass substantially all of their business operations. Each facility is controlled, however, not by HMCA, but by the physician owner, or in the case of the six Florida sites owned by HMCA subsidiaries, by the medical director. All medical services are performed by physicians and other medical personnel under the physician-owner’s supervision. HMCA is the management company and performs services of a non-medical nature. These services include:
1. Offices and Equipment. HMCA identifies, negotiates leases for and/or provides office space and equipment to its clients. This includes technologically sophisticated medical equipment. HMCA also provides improvements to leaseholds, assistance in site selection and advice on improving, updating, expanding and adapting to new technology.
2. Personnel. HMCA staffs all the non-medical positions of its clients with its own employees, eliminating the client’s need to interview, train and manage non-medical employees. HMCA processes the necessary tax, insurance and other documentation relating to employees.
3. Administrative. HMCA assists in the scheduling of patient appointments, purchasing of office and medical supplies and equipment and handling of reporting, accounting, processing and filing systems. It prepares and files the physician portions of complex applications to enable its clients to participate in managed care programs and to qualify for insurance reimbursement. HMCA assists the clients to implement programs and procedures to ensure full and timely regulatory compliance and appropriate cost reimbursement under no-fault insurance and Workers’ Compensation guidelines, as well as compliance with other applicable governmental requirements and regulations, including HIPAA and other privacy requirements.
4. Billing and Collections. HMCA is responsible for the billing and collection of revenues from third-party payors including those governed by No-Fault and Workers’ Compensation statutes.
5. Cost Saving Programs. Based on available volume discounts, HMCA seeks to assist in obtaining favorable pricing for office and medical supplies, medical imaging film, equipment, contrast agents, such as gadolinuim, and magnavist and other inventory for its clients.
6. Diagnostic Imaging and Ancillary Services. HMCA can offer access to diagnostic imaging equipment through diagnostic imaging facilities it manages. The Company is expanding the ancillary services offered in its network to include x-rays, and other MRI equipment such as high-field (1.5 or 3.0 Tesla magnet strength) MRI scanners and extremity MRI scanners.
7. Marketing Strategies. HMCA is responsible for developing and proposing marketing plans for its clients.
8. Expansion Plans. HMCA assists the clients in developing expansion plans including the opening of new or replacement facilities where appropriate.
HMCA’s objective is to free physicians from as many non-medical duties as is practicable, allowing physicians to spend less time on business and administrative matters and more time practicing medicine.
| Page 17 |
FONAR CORPORATION AND SUBSIDIARIES
The exceptions to this general model of operation are six of the facilities located in Florida. These Florida facilities are owned by limited liability companies which, as our subsidiaries, conduct their operations directly and bill and collect their fees from the patients and third-party payors.
The facilities enter into contracts with third-party payors, including managed care companies. None of HMCA’s clients, however, participate in any capitated plans or other risk sharing arrangements. Capitated plans are those HMO programs where the provider is paid a flat monthly fee per patient.
The management fees payable by the facilities to HMCA are flat monthly fees. In fiscal 2023, the aggregate amount of active management fees was $4,860,732 per month. In fiscal 2022, the aggregate amount of active management fees was $4,865,443 per month.
Fees under the management agreements are subject to adjustment by mutual agreement on an annual basis.
Timothy Damadian currently owns three HMCA-managed MRI facilities in Florida. The facilities were owned by Dr. Damadian until his passing in August of 2022. The fees for these three sites are flat monthly fees which are subject to adjustment by mutual agreement on an annual basis. In fiscal 2023 and fiscal 2022, the aggregate monthly amount of management fees payable to HMCA by these sites was $995,825.
The six Florida facilities owned by HMCA subsidiaries directly bill their patients or the patients’ insurance carriers. Patient fees net of provision for bad debts were $29,793,993 in fiscal 2023 as compared to $29,582,238 in fiscal 2022.
HMCA had previously contracted with an outside billing company (located in Melville, New York) which performed billing and collection for their clients’ No-Fault and Workers’ Compensation business. The Company had entered into a one year renewable agreement to provide IT services to the billing company for a monthly fee of $23,884. This agreement was terminated on May 31, 2023. HMCA has been handling these billing and collection services internally since the termination of this agreement.
HMCA MARKETING
HMCA’s marketing strategy is to expand the business and improve the facilities which it manages. HMCA is seeking to increase the number of locations of those facilities where market conditions are promising and to promote growth of our clients’ and Florida subsidiaries’ patient volume and revenue.
| Page 18 |
FONAR CORPORATION AND SUBSIDIARIES
DIAGNOSTIC IMAGING FACILITIES
Diagnostic imaging facilities managed by HMCA provide diagnostic imaging services to patients referred by physicians. The facilities are operated in a manner which eliminates the admission and other administrative inconveniences of in-hospital diagnostic imaging services. Imaging services are performed in an outpatient setting by trained medical technologists under the direction of physicians. Following diagnostic procedures, the images are reviewed by the interpreting physicians who prepare reports of these tests and their findings. The vast majority of reports for the New York facilities are transcribed by HMCA personnel and the remainder are outsourced to professional transcription services. Reports for the Florida facilities are outsourced to professional transcription services.
HMCA develops marketing programs and educational programs in an effort to establish and maintain referring physician relationships for our clients and Florida subsidiaries.
Managed care providers are an important factor in the diagnostic imaging industry. To further its position, HMCA is seeking to expand the imaging modalities offered at its managed and owned diagnostic imaging facilities. Four facilities in New York and eight facilities in Florida have two or more MRI scanners. One facility in New York and two in Florida also perform X-rays. During fiscal 2023, a new location was opened in Casselberry, Florida.
REIMBURSEMENT
HMCA’s clients receive reimbursements for their services through Medicare, Medicaid, managed care, private commercial insurance, third-party administrators, Workers’ Compensation, No-Fault and other insurance.
Medicare
The Medicare program provides reimbursement for hospitalization, physician, diagnostic and certain other services to eligible persons 65 years of age and over and certain other individuals. Providers are paid by the federal government in accordance with regulations promulgated by the Department of Health and Human Services, HSS, and generally accept the payment with nominal deductible and co-insurance amounts required to be paid by the service recipient, as payment in full. Hospital inpatient services are reimbursed under a prospective payment system. Hospitals receive a specific prospective payment for inpatient treatment services based upon the diagnosis of the patient.
Under Medicare’s prospective payment system for hospital outpatient services, or OPPS, a hospital is paid for outpatient services on a rate per service basis that varies according to the ambulatory payment classification group, or APC, to which the service is assigned rather than on a hospital’s costs. Each year the Centers for Medicare and Medicaid Services, or CMS, publishes new APC rates that are determined in accordance with the promulgated methodology.
Services provided in non-hospital based freestanding facilities are paid under the Medicare Physician Fee Schedule, or MPFS. All of HMCA’s clients are presently in this category. The MPFS is updated on an annual basis and sometimes modified more frequently.
| Page 19 |
FONAR CORPORATION AND SUBSIDIARIES
We have experienced reimbursement reductions for radiology services provided to Medicare beneficiaries, including reductions pursuant to the Deficit Reduction Act, or DRA.
CMS’ 2010 regulatory changes to the MPFS included a downward adjustment to services primarily involving the technical component rather than the physician work component, by adjusting downward malpractice payments for these services. These adjustments have been phased in over a four year period. For our fiscal year ended June 30, 2023, Medicare revenues represented approximately 2.9% of the revenues for HMCA’s clients and subsidiaries as compared to 3.2% for the fiscal year ended June 30, 2022.
Medicaid
The Medicaid program is a jointly-funded federal and state program providing coverage for low-income persons. In addition to federally-mandated basic services, the services offered and reimbursement methods vary from state to state. In many states, Medicaid reimbursement is patterned after the Medicare program; however, an increasing number of states have established or are establishing payment methodologies intended to provide healthcare services to Medicaid patients through managed care arrangements. In fiscal 2023, approximately 0.05% of the revenues of HMCA’s clients were attributable to Medicaid, as compared to 0.07% in fiscal 2022.
Managed Care and Private Insurance
Health Maintenance Organizations, or HMO’s, Preferred Provider Organizations, or PPOs, and other managed care organizations attempt to control the cost of healthcare services by a variety of measures, including imposing lower payment rates, preauthorization requirements, limiting services and mandating less costly treatment alternatives. Managed care contracting is competitive and reimbursement schedules in many cases can be at or below Medicare reimbursement levels. Some managed care organizations have reduced or otherwise limited, and other managed care organizations may reduce or otherwise limit, reimbursement in response to reductions in government reimbursement. These reductions could have an adverse impact on our financial condition and results of operations. These reductions have been, and any future reductions may be, similar to the reimbursement reductions previously proposed.
HMCA COMPETITION
The physician and diagnostic management services field is highly competitive. A number of large hospitals have acquired medical practices and this trend may continue. HMCA expects that more competition will develop. Many competitors have greater financial and other resources than HMCA.
| Page 20 |
FONAR CORPORATION AND SUBSIDIARIES
With respect to the diagnostic imaging facilities managed by HMCA, the outpatient diagnostic imaging industry is highly competitive. Competition focuses primarily on attracting physician referrals at the local market level and increasing referrals through relationships with managed care organizations, as well as emphasizing to potential referral sources the advantages of Upright® MRI scanning. HMCA believes that principal competitors for the diagnostic imaging centers are hospitals and independent or management company-owned imaging centers. Competitive factors include quality and timeliness of test results, ability to develop and maintain relationships with managed care organizations and referring physicians, type and quality of equipment, facility location, convenience of scheduling and availability of patient appointment times. HMCA believes that it will be able to effectively meet the competition in the outpatient diagnostic imaging industry with the FONAR Upright® MRI scanners and strategically placed high field MRI scanners at its facilities.
GOVERNMENT REGULATION APPLICABLE TO HMCA
FEDERAL REGULATION
The healthcare industry is highly regulated and changes in laws and regulations can be significant. Changes in the law or new interpretation of existing laws can have a material effect on our permissible activities, the relative costs associated with doing business and the amount of reimbursement by government and other third-party payors.
Federal False Claims Act
The federal False Claims Act and, in particular, the False Claims Act’s “qui tam” or “whistleblower” provisions allow a private individual to bring actions in the name of the government alleging that a defendant has made false claims for payment from federal funds. After the individual has initiated the lawsuit the government must decide whether to intervene in the lawsuit and to become the primary prosecutor. If the government declines to join the lawsuit, the individual may choose to pursue the case alone, although the government must be kept apprised of the progress of the lawsuit, and may intervene later. Whether or not the federal government intervenes in the case, it will receive the majority of any recovery.
When an entity is determined to have violated the federal False Claims Act, it must pay three times the actual damages sustained by the government, plus mandatory civil penalties for each separate false claim and the government’s attorneys’ fees. Liability arises when an entity knowingly submits, or causes someone else to submit, a false claim for reimbursement to the federal government. The False Claims Act defines the term “knowingly” broadly, though simple negligence will not give rise to liability under the False Claims Act. Examples of the other actions which may lead to liability under the False Claims Act are set forth below:
Failure to comply with the many technical billing requirements applicable to our Medicare and Medicaid business;
Failure to comply with the prohibition against billing for services ordered or supervised by a physician who is excluded from any federal healthcare program, or the prohibition against employing or contracting with any person or entity excluded from any federal healthcare program;
| Page 21 |
FONAR CORPORATION AND SUBSIDIARIES
Failure to comply with the Medicare physician supervision requirements for the services we provide, or the Medicare documentation requirements concerning physician supervision.
The Fraud Enforcement and Recovery Act of 2009 expanded the scope of the False Claims Act by, among other things, broadening protections for whistleblowers and creating liability for knowingly retaining a government overpayment, acting in deliberate ignorance of a government overpayment or acting in reckless disregard of a government overpayment. The healthcare reform bills in the form of the Patient Protection and Affordable Care Act, as amended by the Health Care and Education Reconciliation Act of 2010 (collectively, “PPACA”) expanded on changes made by the 2009 Fraud Enforcement and Recovery Act with regard to such “reverse false claims.” Under PPACA, the knowing failure to report and return an overpayment within 60 days of identifying the overpayment or by the date a corresponding cost report is due, whichever is later, constitutes a violation of the False Claims Act. HMCA and its clients have never been sued under the False Claims Act and believe they are in compliance with the law.
Stark Law
Under the federal Self-Referral Law, also referred to as the “Stark Law”, which is applicable to Medicare and Medicaid patients, and the self-referral laws of various States, certain health practitioners, including physicians, chiropractors and podiatrists, are prohibited from referring their patients for the provision of designated health services, including diagnostic imaging and physical therapy services, to any entity with which they or their immediate family members have a financial relationship, unless the referral fits within one of the specific exceptions in the statutes or regulations. The federal government has taken the position that a violation of the federal Stark Law is also a violation of the Federal False Claims Act. Statutory exceptions under the Stark Law include, among others, direct physician services, in-office ancillary services rendered within a group practice, space and equipment rental and services rendered to enrollees of certain prepaid health plans. Some of these exceptions are also available under the State self-referral laws. HMCA believes that it and its clients are in compliance with these laws.
Anti-kickback Regulation
We are subject to federal and state laws which govern financial and other arrangements between healthcare providers. These include the federal anti-kickback statute which, among other things, prohibits the knowing and willful solicitation, offer, payment or receipt of any remuneration, direct or indirect, in cash or in kind, in return for or to induce the referral of patients for items or services covered by Medicare, Medicaid and certain other governmental health programs. Under PPACA, knowledge of the anti-kickback statute or the specific intent to violate the law is not required. Violation of the anti-kickback statute may result in civil or criminal penalties and exclusion from the Medicare, Medicaid and other federal healthcare programs, and according to PPACA, now provides a basis for liability under the False Claims Act. In addition, it is possible that private parties may file “qui tam” actions based on claims resulting from relationships that violate the anti-kickback statute, seeking significant financial rewards. Many states have enacted similar statutes, which are not limited to items and services paid for under Medicare or a federally funded healthcare program. Neither HMCA nor its clients engage in this practice.
| Page 22 |
FONAR CORPORATION AND SUBSIDIARIES
In fiscal 2023, approximately 2.9% of the revenues of HMCA’s clients were attributable to Medicare and 0.05% were attributable to Medicaid. In fiscal 2022, approximately 3.2% of the revenues of HMCA’s clients were attributable to Medicare and 0.07% were attributable to Medicaid.
Deficit Reduction Act (DRA)
On February 8, 2006, the President signed into law the DRA. Effective January 1, 2007, the DRA provides that Medicare reimbursement for the technical component for imaging services (excluding diagnostic and screening mammography) performed in freestanding facilities will be capped. Payment is the lesser of the Medicare Physician Fee Schedule or the Hospital Outpatient Prospective Payment System (OPPS) rates. Implementation of these reimbursement reductions contained in the DRA has had an adverse effect on our business. We have been able to counter this effect by increasing our clients’ scan volumes through our vigorous marketing efforts and reducing our operating expenses.
The DRA also codified the reduction in reimbursement for multiple images on contiguous body parts previously announced by CMS, the agency responsible for administering the Medicare program. In November 2005, CMS announced that it would pay 100% of the technical component of the higher priced imaging procedure and 50% of the technical component of each additional imaging procedure for imaging procedures involving contiguous body parts within a family of codes when performed in the same session. CMS had indicated that it would phase in this 50% rate reduction over two years, so that the reduction was 25% for each additional imaging procedure in 2006 and another 25% reduction in 2007. However, for services furnished on or after July 1, 2010, the PPACA requires the full 50% reduction to be implemented.
Health Insurance Portability and Accountability Act
Congress enacted the Health Insurance Portability and Accountability Act of 1996, or HIPAA, in part, to combat healthcare fraud and to protect the privacy and security of patients’ individually identifiable healthcare information. HIPAA, among other things, amends existing crimes and criminal penalties for Medicare fraud and enacts new federal healthcare fraud crimes, including actions affecting non-governmental healthcare benefit programs by means of false or fraudulent representations in connection with the delivery of healthcare services is subject to a fine or imprisonment, or potentially both. In addition, HIPAA authorizes the imposition of civil money penalties against entities that employ or enter into contracts with excluded Medicare or Medicaid program participants if such entities provide services to federal health program beneficiaries. A finding of liability under HIPAA could have a material adverse effect on our business, financial condition and results of operations.
| Page 23 |
FONAR CORPORATION AND SUBSIDIARIES
Further, HIPAA requires healthcare providers and their business associates to maintain the privacy and security of individually identifiable protected health information (“PHI”). HIPAA imposes federal standards for electronic transactions, for the security of electronic health information and for protecting the privacy of PHI. The Health Information Technology for Economic and Clinical Health Act of 2009 (“HITECH”), signed into law on February 17, 2009, dramatically expanded, among other things, (1) the scope of HIPAA to now apply directly to “business associates,” or independent contractors who receive or obtain PHI in connection with providing a service to a covered entity, (2) substantive security and privacy obligations, including new federal security breach notification requirements to affected individuals, DHHS and prominent media outlets, of certain breaches of unsecured PHI, (3) restrictions on marketing communications and a prohibition on covered entities or business associates from receiving remuneration in exchange for PHI, and (4) the civil and criminal penalties that may be imposed for HIPAA violations, increasing the annual cap in penalties from $25,000 to $1.5 million per occurrence. In 2013 additional legal requirements were adopted to provide further protection for PHI.
In addition, many states have enacted comparable privacy and security statues or regulations that, in some cases, are most stringent than HIPAA requirements. In those cases it may be necessary to modify our operations and procedures to comply with the more stringent state laws, which may entail significant and costly changes for us. We believe that we are in compliance with such state laws and regulations. However, if we fail to comply with applicable state laws and regulations, we could be subject to sanctions.
We believe that we are in compliance with the current HIPAA requirements, as amended by HITECH, together with other legislation and regulations, and comparable state laws, but we anticipate that we may encounter certain costs associated with future compliance. Moreover, we cannot guarantee that enforcement agencies or courts will not make interpretations of the HIPAA standards that are inconsistent with ours, or the interpretations of our contracted radiology practices or their affiliated physicians. A finding of liability under the HIPAA standards may result in significant criminal and civil penalties. Noncompliance also may result in exclusion from participation in government programs, including Medicare and Medicaid. These actions could have a material adverse effect on our business, financial condition, and results of operations.
Civil Money Penalty Law and Other Federal Statutes
The Civil Money Penalty, or CMP, law covers a variety of practices. It provides a means of administrative enforcement of the anti-kickback statute, and prohibits false claims, claims for medically unnecessary services, violations of Medicare participating provider or assignment agreements and other practices. The statute gives the Office of Inspector General of the HHS the power to seek substantial civil fines, exclusion and other sanctions against providers or others who violate the CMP prohibitions.
In addition, in 1996, Congress created a new federal crime: healthcare fraud and false statements relating to healthcare matters. The healthcare fraud statute prohibits knowingly and willfully executing a scheme to defraud any healthcare benefit program, including private payors. A violation of this statute is a felony and may result in fines, imprisonment or exclusion from government sponsored programs such as the Medicare and Medicaid programs.
| Page 24 |
FONAR CORPORATION AND SUBSIDIARIES
Certificates of Need
Some states require hospitals and certain other healthcare facilities and providers to obtain a certificate of need, or CON, or similar regulatory approval prior to establishing certain healthcare operations or services, incurring certain capital projects and/or the acquisition of major medical equipment including MRI and PET/CT systems. We are not operating in any such states.
Patient Protection and Affordable Care Act
On March 23, 2010, President Obama signed into law healthcare reform legislation in the form of PPACA. The implementation of this law has had a significant impact on the healthcare industry. Most of the provisions of PPACA are being phased in over time and can be conceptualized as a broad framework not only to provide health insurance coverage to millions of Americans, but to fundamentally change the delivery of care by bringing together elements of health information technology, evidence-based medicine, chronic disease management, medical “homes,” care collaboration and shared financial risk in a way that will accelerate industry adoption and change. We are unable to predict the full impact of PPACA at this time primarily due to the previous administration’s efforts to repeal and replace the PPACA, or to utilize executive action to modify the Act’s provisions where possible.
State Regulation
In addition to the federal self-referral law and federal Anti-kickback statute, many States, including those in which HMCA and its clients operate, have their own versions of self-referral and anti-kickback laws. These laws are not limited in their applicability, as are the federal laws, to specific programs. HMCA believes that it and its clients are in compliance with these laws.
Various States prohibit business corporations from practicing medicine. Various States, including New York, also prohibit the sharing of professional fees or fee splitting. Consequently, in New York HMCA leases space and equipment to clients and provides clients with a range of non-medical administrative and managerial services for agreed upon fees. Under Florida law a business entity can bill patients and third-party payors directly if that entity is properly licensed through AHCA. All of the nine facilities in Florida are licensed healthcare clinics through AHCA.
HMCA’s clients and subsidiaries generate revenue from patients covered by no-fault insurance and workers’ compensation programs. For the fiscal year ended June 30, 2023 approximately 58.4% of our clients’ receipts were from patients covered by no-fault insurance and approximately 8.6% of our client’s receipts were from patients covered by workers’ compensation programs. For the fiscal year ended June 30, 2022, approximately 57.7.% of HMCA’s clients’ receipts were from patients covered by no-fault insurance and approximately 8.6% of HMCA’s clients’ receipts were from patients covered by workers’ compensation programs. The foregoing numbers do not include payments from third-party administrators. In the event that changes in these laws alter the fee structures or methods of providing service, or impose additional or different requirements, HMCA could be required to modify its business practices and services in ways that could be more costly to HMCA or in ways that decrease the revenues which HMCA receives from its clients.
| Page 25 |
FONAR CORPORATION AND SUBSIDIARIES
Compliance Program
We maintain a program to monitor compliance with federal and state laws and regulations applicable to the healthcare entities. The compliance program includes the adoption of (i) Standards of Conduct for our employees and affiliates and (ii) a process that specifies how employees, affiliates and others may report regulatory or ethical concerns. We believe that our compliance program meets the relevant standards provided by the Office of Inspector General of the Department of Health and Human Services.
An important part of our compliance program consists of conducting periodic audits of various aspects of our operations and that of the contracted radiology practices. We also assist our clients with educational programs designed to familiarize them with the regulatory requirements and specific elements of our compliance program.
HMCA believes that it and its clients are in compliance with applicable Federal, State and local laws. HMCA does not believe that such laws will have any adverse material effect on its business.
EMPLOYEES
FONAR and HMCA had approximately 561 employees as of September 12, 2023. This total number included employees engaged in production, customer support, research and development, information technology, employees engaged in marketing and sales, billing and collection, legal and compliance matters, as well as transcriptionists, Florida technologists, field service technicians and individuals in various administrative positions. A significant number of employees were employed at the MRI facilities managed or owned by HMCA, primarily in administrative positions.
ITEM 1A. RISK FACTORS
An investment in our securities is subject to various risks, the most significant of which are summarized below.
| 1. | Reduced Reimbursement Rates. Most of our revenues are derived from our scanning center business conducted by HMCA. Our scanning center clients and the Florida facilities owned by HMCA are experiencing lower reimbursement rates from Medicare, other government programs and private insurance companies. To the extent possible, we counter these reductions by increasing scanning volume and controlling operating expenses. Inflation in the cost of both materials and labor have limited our ability to control our costs, negatively impacting our ability to maintain profitability in this business segment. |
| 2. | Inflation
and Increasing Interest Rates. Inflation has drastically increased our costs for both materials
and labor. The Federal Reserve has increased interest rates substantially in an attempt to
control inflation, which in turn has increased the cost of capital. Diagnostic imaging facilities
require significant amounts of capital to operate, particularly in the context of opening
new diagnostic imaging centers. These increased costs make it more difficult to achieve organic
growth and extend the time that a new center takes to achieve profitability. Continued costs
increases, coupled with reduced reimbursement rates, may threaten the profitability of our
current operations and cause the cost of expansion to become prohibitively high. |
| Page 26 |
FONAR CORPORATION AND SUBSIDIARIES
| 3. | Demand for MRI Scanners. The reduced margins have a negative effect on our sales of MRI scanners. With lower revenue projections, prospective customers demand lower prices for scanners. Although the reduced reimbursements may not affect foreign demand, a lower number of sales in the aggregate could reduce economies of scale and consequently, profit margins. |
| 4. | Manufacturing Competition. Many if not most of our competing scanner manufacturers have significantly greater financial resources, production capacity, and other resources than we do. Such competitors would include General Electric, Siemens, Hitachi and Phillips. Although FONAR is the only company which can manufacture and sell the unique Stand-Up® (Upright®) MRI scanner, potential customers must be convinced that the purchase of a FONAR scanner is their best choice. We believe that with time, that objective will be reached, particularly with customers scanning patients having neck, back, knee and various orthopedic issues who would benefit from being scanned in weight-bearing positions. |
| 5. | Dependence on Referrals. HMCA derives substantially all of its revenue, directly or indirectly, from fees charged for the diagnostic imaging services performed at the facilities. We depend on referrals of patients from unaffiliated physicians and other third parties to the facilities we manage or own for the services we perform. If these physicians and other third parties were to reduce the number of patients they refer or discontinue referring patients, scan volumes could decrease, which would reduce our net revenue and operating margins. |
| 6. | Pressure to Control Healthcare Costs. One of the principal objectives of health maintenance organizations and preferred provider organizations is to control the cost of healthcare services. Healthcare providers participating in managed care plans may be required to refer diagnostic imaging tests to certain providers depending on the plan in which a covered patient is enrolled. In addition, managed care contracting has become very competitive. The expansion of health maintenance organizations, preferred provider organizations and other managed care organizations in New York or Florida could have a negative impact on the utilization and pricing of services performed at the facilities HMCA manages or owns to the extent these organizations exert control over patients’ access to diagnostic imaging services, selections of the provider of such services and reimbursement rates for those services. |
| 7. | Scanning Facility Competition. The market for diagnostic imaging services is highly competitive. The facilities we manage or own compete for patients on the basis of reputation, location and the quality of diagnostic imaging services. Groups of radiologists, established hospitals, clinics and other independent organizations that own and operate imaging equipment are the principal competitors. |
| Page 27 |
FONAR CORPORATION AND SUBSIDIARIES
| 8. | Eligibility Changes to Insurance Programs. Due to potential decreased availability of healthcare through private employers, the number of patients who are uninsured or participate in governmental programs may increase. Healthcare reform legislation will increase the participation of individuals in the Medicaid program in states that elect to participate in the expanded Medicaid coverage. A shift in payor mix from managed care and other private payors to government payors or an increase in the number of uninsured patients may result in a reduction in the rates of reimbursement or an increase in uncollectible receivables or uncompensated care, with a corresponding decrease in net revenue. Policies now being offered under various insurance plans are expected to reduce demand for MRI scans as they become less affordable. Changes in the eligibility requirements for governmental programs such as the Medicaid program and state decisions on whether to participate in the expansion of such programs also could increase the number of patients who participate in such programs and the number of uninsured patients. Even for those patients who remain in private insurance plans, changes to those plans could increase patient financial responsibility, resulting in a greater risk of uncollectible receivables. These factors and events could have a material adverse effect on our business, financial condition, and results of operations. |
| 9. | Current and future changes in Florida Insurance Law. On March 24, 2023, Florida Governor Ron DeSantis signed into law House Bill 837. Dubbed the Tort Reform Act, the bill makes sweeping changes to Florida’s negligence laws, including reducing the statute of limitations, barring recovery for plaintiffs who are found to be 50% or greater at fault, and changing the rule of evidence regarding admissibility of the costs of prior and future medical treatment. The bill is viewed as a boon to insurance companies, and is largely aimed at reducing the cost of personal injury lawsuits to insurers operating in Florida’s motor vehicle and general liability markets. The full impact of the bill remains to be seen. Certain provisions of the bill are expected to negatively impact our reimbursement percentage and/or reimbursement rates. We expect that some percentage of our patients who are seeking treatment following motor vehicle accidents will not meet the new 51% threshold, and as a result we expect an increase in the percentage of uncollectible billings from those patients. We are unable to estimate what that percentage might be. |
Further, changes to the evidentiary admissibility rules may lead to a higher percentage of our billings being paid at commercial rates instead of at the presently prevailing PIP schedule, a reduction in reimbursement of approximately 60%. These changes will negatively impact our Florida diagnostic imaging facilities (both those we own and those we manage) with more unpaid bills, and lower reimbursement rates. The full extent of those reductions are unclear at this time.
In addition to the above, Florida legislature continues to propose an outright repeal of Florida’s No-Fault law. SB 586 and its companion statue H 429, again propose a repeal of Personal Injury Protection and replacing it with $25,000 Bodily Injury Coverage and Property Damage Liability Coverage. We cannot predict whether Florida will continue to pursue the repeal of the No-Fault Law in light of the passage of HB 837, and whether such efforts will be successful.
| Page 28 |
FONAR CORPORATION AND SUBSIDIARIES
| 10. | Federal and state privacy and information security laws. We must comply with numerous federal and state laws and regulations governing the collection, dissemination, access, use, security and privacy of PHI, including HIPAA and its implementing privacy and security regulations, as amended by the federal HITECH Act. If we fail to comply with applicable privacy and security laws, regulations and standards, properly maintain the integrity of our data, protect our proprietary rights to our systems, or defend against cybersecurity attacks, our business, reputation, results of operations, financial position and cash flows could be materially and adversely affected. |
Information security risks have significantly increased in recent years because of the proliferation of new technologies, the use of the internet and telecommunications technologies to conduct our operations, and the increased sophistication and activities of organized crime, hackers, terrorists and other external parties, including foreign state agents. Our operations rely on the secure processing, transmission and storage of confidential, proprietary and other information in our computer systems and networks.
| 11. | Other changes in Domestic and Worldwide Economic Conditions. We are subject to risk arising from adverse changes in general domestic and global economic conditions, including recession or economic slowdown and disruption of credit markets. Turbulence and uncertainty in the United States and international markets and economies may adversely affect our liquidity, financial condition, revenues, profitability and business operations generally. |
ITEM 1B. UNRESOLVED STAFF COMMENTS
None.
ITEM 2. PROPERTIES
FONAR and HMCA currently lease approximately 78,000 square feet of office and plant space at its principal offices in Melville, New York. The term of the lease runs through November, 2026. Management believes that the premises will be adequate for its current needs. HMCA also maintains office space for the Facilities owned by its subsidiaries in Florida and for its clients at the clients’ sites in New York and Florida under leases having various terms. HMCA owns the building for the client’s premises in Tallahassee, Florida. The Company received approval from the Suffolk County Industrial Development Agency on February 29, 2016 of a 50% property tax abatement, valued at $440,000, over a 10 year period commencing January, 2017.
ITEM 3. LEGAL PROCEEDINGS.
There are no material legal proceedings threatened or pending against the Company.
ITEM 4. MINE SAFETY DISCLOSURES.
Not Applicable
| Page 29 |
FONAR CORPORATION AND SUBSIDIARIES
PART II
ITEM 5. MARKET FOR REGISTRANT’S COMMON EQUITY AND RELATED STOCKHOLDER MATTERS
Our Common Stock is traded on NASDAQ Capital Markets under the symbol FONR.
On September 12, 2023, we had approximately 996 stockholders of record of our Common Stock, 12 stockholders of record of our Class B Common Stock, 3 stockholders of record of our Class C Common Stock and 1,155 stockholders of record of our Class A Non-voting Preferred Stock.
At the present time, the only class of our securities for which there is a market is the Common Stock.
We currently have a policy of retaining earnings to finance the development and expansion of our business. We expect to continue this policy for the foreseeable future.
Performance Graph
The following graph compares the Company’s cumulative total stockholder return on its Common Stock against industry and broad-market indexes which have been compiled by the Nasdaq Global Index Group. The periods commence on June 28, 2019 for five years and end on June 30, 2023.. The graph assumes $100 is invested in FONAR Common Stock (NASDAQ: FONR), the Nasdaq Composite Total Return (Nasdaq Composite), Nasdaq Health Care Management Services (Nasdaq Health), and Nasdaq Medical Equipment (Nasdaq Equipment). The comparisons in the graph below are based on historical data and are not intended to forecast the possible future performance of the common stock.
| Date | June
28, 2019 |
June 30, 2020 | June 30, 2021 | June 30, 2022 | June 30, 2023 | |||||||||||||||
| FONR Common Stock | $ | 100 | $ | 99 | $ | 82 | $ | 71 | $ | 79 | ||||||||||
| Nasdaq Composite | $ | 100 | $ | 127 | $ | 184 | $ | 141 | $ | 178 | ||||||||||
| Nasdaq Health | $ | 100 | $ | 114 | $ | 58 | $ | 193 | $ | 185 | ||||||||||
| Nasdaq Equipment | $ | 100 | $ | 106 | $ | 54 | $ | 129 | $ | 145 | ||||||||||
]
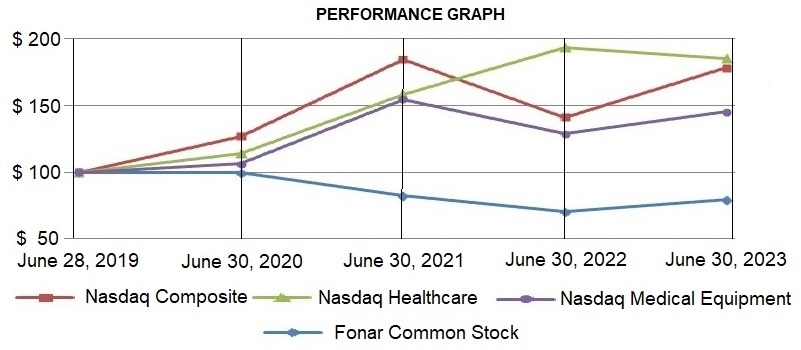
| Page 30 |
FONAR CORPORATION AND SUBSIDIARIES
ITEM 6. [Reserved]
Not applicable.
ITEM 7. MANAGEMENT’S DISCUSSION AND ANALYSIS OF FINANCIAL CONDITION AND RESULTS OF OPERATION.
INTRODUCTION.
FONAR was formed in 1978 to engage in the business of designing, manufacturing and selling MRI scanners. HMCA, a subsidiary of FONAR, provides management services to diagnostic imaging facilities.
FONAR’s principal MRI product is its Upright® MRI (also called Stand-Up® MRI) scanner. The Upright® MRI allows patients to be scanned for the first time under weight-bearing conditions. The Stand-Up® MRI is the only MRI capable of producing images in the weight-bearing state.
At 0.6 Tesla field strength, the Upright® MRI is among the highest field open MRI scanners in the industry, offering non-claustrophobic MRI together with high-field image quality. FONAR’s open MRI scanners were the first high field strength open MRI scanners in the industry.
HMCA generates revenues from providing comprehensive management services, including development, administration, accounting, billing and collection services, together with office space, medical equipment, supplies and non-medical personnel to its clients. Revenues are in the form of fees which are earned under contracts with HMCA’s clients except for its six Florida subsidiaries which engage in the practice of medicine, and bill and collect fees from patients, insurers and other third-party payors directly.
Since March, 2020, the global pandemic of COVID-19 has caused turbulence and uncertainty in the United States and international economies which have adversely affected our workforce, liquidity, financial conditions, revenues, profitability and business operations. The Company has been able to navigate through these challenges and avoid any significant disruption of the business and the volume has risen back to pre-COVID-19 levels. Although we are unable to predict if there will be additional consequences on our operations, the Company believes with the positive cash flows, low debt and cash on hand, it will be able to continue operations going forward.
CRITICAL ACCOUNTING POLICIES
Our discussion and analysis of financial condition and results of operations are based on our consolidated financial statements that were prepared in accordance with U.S. generally accepted accounting principles, or GAAP. Management makes estimates and assumptions when preparing financial statements. These estimates and assumptions affect various matters, including:
Our reported amounts of assets and liabilities in our consolidated balance sheets at the dates of the financial statements;
Our disclosure of contingent assets and liabilities at the dates of the financial statements; and Our reported amounts of net revenue and expenses in our consolidated statements of operations during the reporting periods.
| Page 31 |
FONAR CORPORATION AND SUBSIDIARIES
These estimates involve judgments with respect to numerous factors that are difficult to predict and are beyond management’s control. As a result, actual amounts could differ materially from these estimates.
The Securities and Exchange Commission defines critical accounting estimates as those that are both most important to the portrayal of a company’s financial condition and results of operations and require management’s most difficult, subjective or complex judgment, often as a result of the need to make estimates about the effect of matters that are inherently uncertain and may change in subsequent periods. In the notes to our consolidated financial statements, we discuss our significant accounting policies.
We believe the following critical accounting policies affect our more significant judgments and estimates used in the preparation of our consolidated financial statements. We recognize revenue and related costs of revenue from sales contracts for our MRI scanners and major upgrades, under the percentage-of-completion method. Under this method, we recognize revenue and related costs of revenue, as each sub-assembly is completed. Amounts received in advance of our commencement of production are recorded as customer advances.
We continuously, qualitatively and quantitatively evaluate the realizability (including both positive and negative evidence) of the net deferred tax assets and assess the valuation allowance periodically. Our evaluation considers the financial condition of the Company and both the business conditions and regulatory environment of the industry. If future taxable income or other factors are not consistent with our expectations, an adjustment to our allowance for net deferred tax assets may be required. For net deferred tax assets we consider estimates of future taxable income, including tax planning strategies, in determining whether our net deferred tax assets are more likely than not to be realized. Our ability to project future taxable income may be significantly affected by our ability to determine the impact of regulatory changes which could adversely affect our future profits. As a result, the benefits of our net operating loss carry forwards could expire before they are utilized.
At June 30, 2022, the net deferred tax asset was valued at $12,842,478. At June 30, 2023, the net deferred tax asset was valued at $10,041,960.
We depreciate our long-lived assets over their estimated economic useful lives with the exception of leasehold improvements where we use the shorter of the assets useful lives or the lease term of the facility for which these assets are associated.
The Company provides for medical receivables that could become uncollectible by establishing an allowance for doubtful accounts in order to adjust medical receivables to estimated net realizable value. In evaluating the collectability of medical receivables, the Company considers a number of factors, including the age of the account, historical collection experiences, payor type, current economic conditions and other relevant factors. There are various factors that impact collection trends, such as payor mix, changes in the economy, increase burden on copayments to be made by patients with insurance and business practices related to collection efforts. These factors continuously change and can have an impact on collection trends and the estimation process.
| Page 32 |
FONAR CORPORATION AND SUBSIDIARIES
We amortize our intangible assets, including patents, and capitalized software development costs, over the shorter of the contractual/legal life or the estimated economic life. Our amortization life for patents and capitalized software development costs is 15 to 17 years and 5 years, respectively. Our amortization of the non-competition agreements entered into with certain individuals in connection with the HDM transaction are depreciated over seven years, and customer relationships are amortized over 20 years.
Goodwill is recorded as a result of business combinations. Management evaluates goodwill, at a minimum, on an annual basis and whenever events and changes in circumstances suggest that the carrying amount may not be recoverable. Impairment of goodwill is tested by comparing the reporting unit’s carrying amount, including goodwill, to the fair value of the reporting unit. The fair value of a reporting unit is estimated using a combination of the income or discounted cash flows approach and the market approach, which uses comparable market data. If the carrying amount of the reporting unit exceeds its fair value, goodwill is considered impaired and a second step is performed to measure the amount of impairment loss, if any. Based on our test for goodwill impairment, we noted no impairment related to goodwill. However, if estimates or the related assumptions change in the future, we may be required to record impairment charges to reduce the carrying amount of goodwill.
We periodically assess the recoverability of long-lived assets, including property and equipment, intangibles and management agreements, when there are indications of potential impairment, based on estimates of undiscounted future cash flows. The amount of impairment is calculated by comparing anticipated discounted future cash flows with the carrying value of the related asset. In performing this analysis, management considers such factors as current results, trends, and future prospects, in addition to other economic factors.
RESULTS OF OPERATIONS. FISCAL 2023 COMPARED TO FISCAL 2022
In fiscal 2023, we recognized net income of $12.1 million on revenues of $98.6 million, as compared to net income of $17.2 million on revenues of $97.6 million for fiscal 2022. This represents an increase in revenues of 1.1%. Total costs and expenses increased by 10.9%. Our consolidated operating results decreased by 32.8% to an operating income of $14.8 million for fiscal 2023 as compared to operating income of $22.0 million for fiscal 2022.
Discussion of Operating Results of Medical Equipment Segment
Fiscal 2023 Compared to Fiscal 2022
Revenues attributable to our medical equipment segment increased by 0.5% to $8.3 million in fiscal 2023 from $8.2 million in fiscal 2022, with product sales revenues increasing by 41.2% from $518,000 in fiscal 2022 to $732,000 in fiscal 2023. Service revenue decreased by 2.2% from $7.7 million in fiscal 2022 to $7.5 million in fiscal 2023.
Product sales to unrelated parties increased by 41.2% from $518,000 in fiscal 2022 to $732,000 in fiscal 2023. There were no product sales to related parties in fiscal 2023 or 2022.
| Page 33 |
FONAR CORPORATION AND SUBSIDIARIES
We believe that one of our principal challenges in achieving greater market penetration is attributable to the better name recognition and larger sales forces of our larger competitors such as General Electric, Siemens, Hitachi, Philips and Toshiba.
In addition, lower reimbursement rates have reduced the demand for our MRI products, resulting in lower sales volumes. As a result of fewer sales, service revenues have decreased since as older scanners are taken out of service, there are fewer new scanners available to sign service contracts.
The operating loss for the medical equipment segment increased from an operating loss of $4.6 million in fiscal 2022 to an operating loss of $5.9 million in fiscal 2023. The losses are attributable most significantly to the fact that costs increased by a greater amount than revenues. The increase in costs was primarily due to the increase in business activity which resulted in our increased revenues.
Research and development expenses increased to $1.6 million in fiscal 2023 from $1.5 million in fiscal 2022. Our expenses for fiscal 2023 represented continued research and development of various upgrades for the Upright® MRI scanner.
Discussion of Operating Results of Physician and Diagnostic Services Management Segment
Fiscal 2023 Compared to Fiscal 2022
Revenues attributable to the Company’s physician and diagnostic services management segment, HMCA, increased to $90.4 million in fiscal 2023 as compared to $89.4 million in fiscal 2022. The increase in revenues was due to an increase of $212,000 of patient fees (net of contractual allowances and discounts less provision for bad debts) from patient and third-party payors recognized by six of the facilities in Florida. Management and other fees increased by $799,000.
Cost of revenues as a percentage of the related revenues for our physician and diagnostic services management segment increased from $47.1 million or 52.7% of related revenues for the year ended June 30, 2022 to $49.0 million, or 54.1% of related revenues for the year ended June 30, 2023. The cost relating to these revenues increased more than the revenues.
Operating results of this segment decreased from operating income of $26.6 million in fiscal 2022 to operating income of $20.7 million in fiscal 2023. The decrease is due mainly to more reserves being taken on management fees. We believe that our efforts to expand and improve the operation of our physician and diagnostic services management segment are directly responsible for the profitability of this segment and our company as a whole.
For the fiscal year ended June 30, 2023, 12.1% of total revenues were derived from contract with facilities that were owned by Dr. Raymond V. Damadian until his passing, and currently owned by Timothy Damadian, the Chief Executive Officer of FONAR. 11.8% of total revenues were derived from these contracts for the 2022 fiscal year. The agreements with these MRI facilities are for one-year terms which renew automatically on an annual basis, unless terminated. The fees for these sites, which are located in Florida, are flat monthly fees.
| Page 34 |
FONAR CORPORATION AND SUBSIDIARIES
Discussion of Certain Consolidated Results of Operations
Fiscal 2023 Compared to Fiscal 2022
Interest and investment income increased in 2023 compared to 2022. We recognized interest income of $1.2 million in 2023 as compared to $247,158 in fiscal 2022, representing an increase of 394.4%. This is due to the increase in the prime interest rate and the Company placing cash in interest bearing accounts.
Interest expense of $50,131 was recognized in fiscal 2023, as compared to interest expense of $346,552 in fiscal 2022. The decrease in interest expense is attributable to an assessment of additional taxes and interest in connection with a state income tax audit in fiscal 2022.
The 29.2% noncontrolling interest allocations of $2,751,000 and $4,793,000 for fiscal 2023 and fiscal 2022, respectively, have been calculated by Income from operations, and adding depreciation and amortization net of miscellaneous losses and other income from the Physician and Diagnostic Service Management segment (See Note 16).
While revenue increased by 1.1% selling, general and administrative expenses increased by 25.0% to $29.4 million in fiscal 2023 from $23.5 million in fiscal 2022. This increase in selling, general and administrative expenses was due to placing more reserves on management fees and other receivables and from the impact of the COVID-19 virus as compared to fiscal 2022. It is too early to know how much of these reserves will be recovered. FONAR also resolved certain sales tax liabilities during the year and was able to reverse accrued interest and penalties of $55,000 and $119,000 for fiscal years ending 2023 and 2022 respectively which was recorded under selling, general and administrative expenses in the prior year.
Revenue from service and repair fees decreased from $7.7 million in fiscal 2022 to $7.5 million in fiscal 2023.
Continuing our tradition as the originator of MRI, we remain committed to maintaining our position as the leading innovator of the industry through investing in research and development. In fiscal 2023 we continued our investment in the development of various upgrades for the UPRIGHT® MRI, with an investment of $1,567,749 in research and development, none of which was capitalized, as compared to $1,494,181, none of which was capitalized, in fiscal 2022. The research and development expenditures were approximately 18.9% of revenues attributable to our medical equipment segment and 1.5% of total revenues in 2023, and 18.2% of medical equipment segment revenues and 1.5% of total revenues in fiscal 2022. This represented a 4.9% increase in research and development expenditures in fiscal 2023 as compared to fiscal 2022.
For the physician and diagnostic services management segment, HMCA, revenues increased to $90.4 million in fiscal 2023 as compared to $89.3 million in fiscal 2022. This is primarily attributable to an increase in patient scans resulting from our marketing efforts.
| Page 35 |
FONAR CORPORATION AND SUBSIDIARIES
For the fiscal year 2023 the Company recorded an income tax expense of $3.6 million compared with an income tax expense of $5.5 million for 2022. The income tax benefits are attributable to the expected tax benefits associated with the projected realization and utilization of our net operating losses in future periods. The Company has recorded a deferred tax asset of $10.0 million as of June 30, 2023, primarily relating to the tax benefits from the net operating loss carry forwards, allowance for doubtful accounts and tax credits available to offset future taxable income. The utilization of these tax benefits is dependent on the Company generating future taxable income and other factors. A partial valuation allowance will be maintained until evidence exists to support that it is no longer needed, (principally related to research and development credits). Although the Company is expecting to generate taxable income in future periods, we cannot accurately measure the full impact of the adoption of healthcare regulations, including the impact of continuing changes in MRI scanning reimbursement rates, which could materially impact operations. A partial valuation allowance will be maintained until evidence exists to support that it is no longer needed. As of June 30, 2023, the valuation allowance was $364,000.
We have been taking steps to improve HMCA revenues by our marketing efforts, which focus on the unique capability of our Upright® MRI scanners to scan patients in different positions. We have also been increasing the number of health insurance plans in which our clients participate.
Our management fees are dependent on collection by our clients of fees from reimbursements from Medicare, Medicaid, private insurance, no fault and workers’ compensation carriers, self–pay and other third-party payors. The health care industry is experiencing the effects of the federal and state governments’ trend toward cost containment, as governments and other third-party payors seek to impose lower reimbursement and utilization rates and negotiate reduced payment schedules with providers. The cost-containment measures, consolidated with the increasing influence of managed-care payors and competition for patients, have resulted in reduced rates of reimbursement for services provided by our clients from time to time. Our future revenues and results of operations may be adversely impacted by future reductions in reimbursement rates.
Certain third-party payors have proposed and implemented changes in the methods and rates of reimbursement that have had the effect of substantially decreasing reimbursement for diagnostic imaging services that HMCA’s clients provide. To the extent reimbursement from third-party payors is reduced, it will likely have an adverse impact on the rates they pay us, as they would need to reduce the management fees they pay HMCA to offset such decreased reimbursement rates. Furthermore, many commercial health care insurance arrangements are changing, so that individuals bear greater financial responsibility through high deductible plans, co-insurance and higher co-payments, which may result in patients delaying or foregoing medical procedures. More frequently, however, patients are scanned and we experience difficulty in collecting deductibles and co-payments. We expect recent changes to the Florida insurance laws to result in less patients being reimbursed through no-fault auto insurance, resulting in both lower reimbursement rates and a higher rate of uncollectible billings. We expect that any further changes to the rates or methods of reimbursement for services, which reduce the reimbursement per scan of our clients may partially offset the increases in scan volume we are working to achieve for our clients, and indirectly will result in a decline in our revenues.
| Page 36 |
FONAR CORPORATION AND SUBSIDIARIES
In addition, the use of radiology benefit managers, or RBM’s has increased in recent years. It is common practice for health insurance carriers to contract with RBMs to manage utilization of diagnostic imaging procedures for their insureds. In many cases, this leads to lower utilization of imaging procedures based on a determination of medical necessity. The efficacy of RBMs is still a highly controversial topic. We cannot predict whether the use of RBMs will negatively impact our business, but it is possible that our financial position and results of operations could be negatively affected.
LIQUIDITY AND CAPITAL RESOURCES
Cash, and cash equivalents increased by 5.2% from $48.7 million at June 30, 2022 to $51.3 million at June 30, 2023.
Cash provided by operating activities for fiscal 2023 approximated $14.5 million. Cash provided by operating activities was attributable to the net income of $12.1 million, depreciation and amortization of $4.5 million, provision for bad debts of $5.5 million, deferred income tax expense benefit of $3.0 million which was offset by the increase in accounts, and medical and management fee receivables of $8.1 million.
Cash used in investing activities for fiscal 2023 approximated $4.3 million. The cash used in investing activities was attributable to purchases of property and equipment of $4.2 million and costs of patents of $120,000.
Cash used in financing activities for fiscal 2023 approximated $7.6 million. The principal uses of cash used in financing activities included the repayment of borrowings and capital lease obligations of $37,000, purchase of treasury stock of $1.8 million and distributions to non-controlling interests of $5.8 million.
Total liabilities decreased by 6.3% during fiscal 2023, from approximately $53.1 million at June 30, 2022 to approximately $49.8 million at June 30, 2023.
At June 30, 2023, we had working capital of approximately $110.0 million as compared to working capital of $101.9 million at June 30, 2022, and stockholders’ equity of $150.8 million at June 30, 2023 as compared to stockholders’ equity of $146.2 million at June 30, 2022. For the year ended June 30, 2023, we realized a net income of $12.1 million.
Our principal sources of liquidity are derived from revenues.
Our business plan includes a program for manufacturing and selling our Upright® MRI scanners. In addition, we are enhancing our revenue by participating in the physician and diagnostic services management business through our subsidiary, HMCA and have upgraded the facilities which it manages, most significantly by the replacement of the original MRI scanners with new Upright® MRI scanners. As of June 30, 2023, HMCA manages a total of 41 MRI scanners of which 24 MRI scanners are located in New York and 17 are located in Florida. We have also intensified our marketing activities through the hiring of additional marketers for HMCA’s clients.
| Page 37 |
FONAR CORPORATION AND SUBSIDIARIES
Our business plan also calls for a continuing emphasis on providing our customers with enhanced equipment service and maintenance capabilities and delivering state-of-the-art, innovative and high quality equipment upgrades at competitive prices. Fees for on-going service and maintenance from our installed base of scanners were $7.7 million for the year ended June 30, 2022 and $7.5 million for the year ended June 30, 2023.
In order to promote profitability and to reduce demands on our cash and other liquid reserves, we maintain an aggressive program of cost containment. Previously, these measures included consolidating HMCA’s office space with FONAR’s office space and reducing the size of our workforce, compensation and benefits. We continue to attempt to contain expenses across the board, despite significant increases in the cost of labor and materials as the result of inflation. The cost control efforts are intended to enable us to withstand periods of low volumes of MRI scanner sales, by keeping expenditures at levels which can be supported by service revenues and HMCA revenues.
Current economic credit conditions have contributed to a slower than optimal business environment. As a result our business may suffer, should the credit markets not improve in the near future. The direct impact of these conditions is not fully known.
Revenues from HMCA have been the principal reason for our profitability, and we have so far been able to maintain and increase such revenues by increasing the number of scans being performed by the sites we manage and those we own, notwithstanding reductions in reimbursement rates from third-party payors. The likelihood and effect of any subsequent reductions is not fully known.
Capital expenditures for fiscal 2023 approximated $4.3 million. Capitalized patent costs were approximately $120,000. Purchases of property and equipment were approximately $4.2 million.
FONAR is has not committed to making any material capital expenditures in the 2024 fiscal year.
The Company believes that its business plan has been responsible for the past five consecutive fiscal years of profitability (fiscal 2023, fiscal 2022, fiscal 2021, fiscal 2020 and fiscal 2019) and that its capital resources will be adequate to support operations at current levels through September 30, 2024.
On September 13, 2022, the Company adopted a stock repurchase plan. On September 26, 2022, the Board of Directors has approved up to $9 million to be repurchased under the plan which will be purchased on the open market at prevailing prices. During fiscal 2023, we repurchased 103,148 shares for $1.8 million.
During August 2023 the Company renewed their revolving credit agreement. The terms include borrowing limits of up to $10,000,000 and the agreement was extended to November 15, 2023. The interest rate on unpaid principal remains at 4% along with certain financial covenants still applicable.
| Page 38 |
FONAR CORPORATION AND SUBSIDIARIES
ITEM 7A. QUALITATIVE AND QUANTITATIVE DISCLOSURES ABOUT MARKET RISK
The Company does not have any investments in marketable securities, foreign currencies, mutual funds, certificates of deposit or other fixed rate instruments. All of our funds are in cash accounts or money market accounts which are liquid.
All of our revenue, expense and capital purchasing activities are transacted in United States dollars.
See Note 10 to the consolidated Financial Statements for information on long-term debt.
ITEM 8.
FINANCIAL STATEMENTS
INDEX TO CONSOLIDATED FINANCIAL STATEMENTS
| PAGE. | |
| REPORT OF INDEPENDENT REGISTERED PUBLIC ACCOUNTING FIRM | 40 |
| CONSOLIDATED BALANCE SHEETS | 43 |
| At June 30, 2023 and 2022 | |
| CONSOLIDATED STATEMENTS OF INCOME | 46 |
| For the Years Ended June 30, 2023 and 2022 | |
| CONSOLIDATED STATEMENTS OF STOCKHOLDERS’ EQUITY | 48 |
| For the Years Ended June 30, 2023 and 2022 | |
| CONSOLIDATED STATEMENTS OF CASH FLOWS | 50 |
| For the Years Ended June 30, 2023 and 2022 | |
| NOTES TO CONSOLIDATED FINANCIAL STATEMENTS | 52 |
| Page 39 |
REPORT OF INDEPENDENT REGISTERED PUBLIC ACCOUNTING FIRM
To the Shareholders and Board of Directors of
FONAR Corporation and Subsidiaries
Opinion on the Financial Statements
We have audited the accompanying consolidated balance sheets of FONAR Corporation and Subsidiaries (the “Company”) as of June 30, 2023 and 2022, the related consolidated statements of income, stockholders’ equity and cash flows for each of the two years in the period ended June 30, 2023, and the related notes (collectively referred to as the “financial statements”). In our opinion, the financial statements present fairly, in all material respects, the financial position of the Company as of June 30, 2023 and 2022, and the results of its operations and its cash flows for each of the two years in the period ended June 30, 2023, in conformity with accounting principles generally accepted in the United States of America.
Basis for Opinion
These financial statements are the responsibility of the Company's management. Our responsibility is to express an opinion on the Company's financial statements based on our audit. We are a public accounting firm registered with the Public Company Accounting Oversight Board (United States) ("PCAOB") and are required to be independent with respect to the Company in accordance with the U.S. federal securities laws and the applicable rules and regulations of the Securities and Exchange Commission and the PCAOB.
We conducted our audits in accordance with the standards of the PCAOB. Those standards require that we plan and perform the audits to obtain reasonable assurance about whether the financial statements are free of material misstatement, whether due to error or fraud. The Company is not required to have, nor were we engaged to perform, an audit of its internal control over financial reporting. As part of our audits we are required to obtain an understanding of internal control over financial reporting but not for the purpose of expressing an opinion on the effectiveness of the Company's internal control over financial reporting. Accordingly, we express no such opinion.
Our audits included performing procedures to assess the risks of material misstatement of the financial statements, whether due to error or fraud, and performing procedures that respond to those risks. Such procedures included examining, on a test basis, evidence regarding the amounts and disclosures in the financial statements. Our audits also included evaluating the accounting principles used and significant estimates made by management, as well as evaluating the overall presentation of the financial statements. We believe that our audits provide a reasonable basis for our opinion.
| Page 40 |
REPORT OF INDEPENDENT REGISTERED PUBLIC ACCOUNTING FIRM (Continued)
Critical Audit Matters
The critical audit matters communicated below are matters arising from the current period audit of the financial statements that were communicated or required to be communicated to the audit committee and that: (1) relate to accounts or disclosures that are material to the financial statements and (2) involved our especially challenging, subjective, or complex judgments. The communication of critical audit matters does not alter in any way our opinion on the financial statements, taken as a whole, and we are not, by communicating the critical audit matters below, providing separate opinions on the critical audit matters or on the accounts or disclosures to which they relate.
Medical Accounts Receivable Reserve – Refer to Note 3 to the financial statements
Critical Audit Matter Description
Medical accounts receivable is recorded at net realizable value based on the estimated amounts the Company expects to receive from patients and third-party payers. Estimates of contractual allowances under managed care, commercial, and governmental insurance plans are based upon the payment terms specified in the related contractual agreements or as mandated under government payer programs. Management continually reviews the contractual allowance estimation process to consider and incorporate updates to laws and regulations and the frequent changes in managed care contractual terms resulting from contract renegotiations and renewals. Receivables related to uninsured patients and uninsured copayment and deductible amounts for patients who have health insurance coverage may have discounts applied. The Company also records estimated implicit price concessions (based on historical experience) related to accounts to record the accounts receivable at the amount the Company expects to collect from patients and third-party payers. This implied concession requires extensive judgment and subjective assumptions. Implicit price concessions relate primarily to amounts due directly from patients and are based upon management’s assessment of historical write-offs and expected net collections, business and economic conditions, trends in federal, state, and private employer health care coverage, and other collection indicators. Auditing management’s estimate of the price concessions was complex and judgmental due to the significant data inputs and subjective assumptions utilized in determining the net realizable value of accounts receivable.
How the Critical Audit Matter Was Addressed in the Audit
Our audit procedures related to the net realizable value of patient accounts receivable included the following:
| - | We obtained an understanding, evaluated the design, and tested the operating effectiveness of certain controls that address the risks of material misstatement relating to the measurement of service fee revenue and receivables. |
| - | We tested informational technology general controls around the Company’s billing system and associated database. |
| - | We evaluated management’s methodology and related assumptions, including cash collections, by comparing actual results to management’s historical estimates. |
| Page 41 |
REPORT OF INDEPENDENT REGISTERED PUBLIC ACCOUNTING FIRM (Continued)
| - | We tested the underlying data related to the recognition of patient level charges and the subsequent activities, including cash collections and non-cash adjustments. |
| - | We tested the contractual rates set forth by the third-party payers which are input into the Company’s billing system and then billed to patients and/or third-party payers. |
| - | We tested the mathematical accuracy of the estimates applied to period-end accounts receivable. |
| - | We evaluated the appropriateness of the industry, economic, and Company factors that were used in determining the net realizable value of patient accounts receivable. |
Management Fee Accounts Receivable Reserve – Refer to Note 3 to the financial statements.
Management fee accounts receivable is related to fees outstanding from the related and non-related professional corporations (“PCs”) under management agreements. Payment of the outstanding fees is dependent on the PCs ability to collect fees from third-party payers and patients because the management fees are collateralized by the PCs accounts receivable. The Company records the management fee accounts receivables net of the estimated implicit price concessions based on the PCs likelihood to collect on the accounts. Implicit price concessions on the PCs are estimated by management in the same manner the patient accounts receivable are analyzed. This implied concession requires extensive judgment and subjective assumptions. Implicit price concessions relate primarily to amounts due directly from patients and are based upon management’s assessment of historical write-offs and expected net collections, business and economic conditions, trends in federal, state, and private employer health care coverage, and other collection indicators. Auditing management’s estimate of the price concessions was complex and judgmental due to the significant data inputs and subjective assumptions utilized in determining the net realizable value of accounts receivable.
How the Critical Audit Matter Was Addressed in the Audit
Our audit procedures related to the management fee accounts receivable reserve are consistent with the audit procedures associated with the patient fee accounts receivable reserve. In addition, we traced the management fees to the underlying agreements and the general ledger.
/s/ Marcum llp
We have served as the Company’s auditor since 1990, such date takes into account the merger of Tabb, Conigliaro, McGann, P.C. (“Tabb”) into another firm in approximately 2001 and the former partners of Tabb joining Marcum LLP in 2002.
September 28, 2023
| Page 42 |
FONAR CORPORATION AND SUBSIDIARIES
CONSOLIDATED BALANCE SHEETS
ASSETS
| June 30, | ||||||||
| 2023 | 2022 | |||||||
| Current Assets: | ||||||||
| Cash and cash equivalents | $ | $ | ||||||
| Short-term investments | ||||||||
| Accounts
receivable – net of allowances for doubtful accounts of $ | ||||||||
| Medical receivables – net | ||||||||
| Management
and other fees receivable – net of allowances for doubtful accounts of $ | ||||||||
| Management
and other fees receivable – related party medical practices – net of allowances for doubtful accounts of $ | ||||||||
| Inventories | ||||||||
| Prepaid expenses and other current assets | ||||||||
| Total Current Assets | ||||||||
| Accounts receivable – long term | ||||||||
| Deferred income tax asset | ||||||||
| Property and equipment – net | ||||||||
| Right-of-use-asset – operating leases | ||||||||
| Right-of-use-asset – financing lease | ||||||||
| Goodwill | ||||||||
| Other intangible assets – net | ||||||||
| Other assets | ||||||||
| Total Assets | $ | $ | ||||||
See accompanying notes to consolidated financial statements.
| Page 43 |
FONAR CORPORATION AND SUBSIDIARIES
CONSOLIDATED BALANCE SHEETS
LIABILITIES
| June 30, | ||||||||
| 2023 | 2022 | |||||||
| Current Liabilities: | ||||||||
| Current portion of long-term debt | $ | $ | ||||||
| Accounts payable | ||||||||
| Other current liabilities | ||||||||
| Operating lease liability – current portion | ||||||||
| Financing lease liability – current portion | ||||||||
| Unearned revenue on service contracts | ||||||||
| Customer deposits | ||||||||
| Total Current Liabilities | ||||||||
| Long-Term Liabilities: | ||||||||
| Unearned revenue on service contracts | ||||||||
| Deferred income tax liability | ||||||||
| Due to related party medical practices | ||||||||
| Operating lease liability – net of current portion | ||||||||
| Financing lease liability – net of current portion | ||||||||
| Long-term debt and capital leases, less current portion | ||||||||
| Other liabilities | ||||||||
| Total Long-Term Liabilities | ||||||||
| Total Liabilities | ||||||||
Commitments, Contingencies and Other Matters
See accompanying notes to consolidated financial statements.
| Page 44 |
FONAR CORPORATION AND SUBSIDIARIES
CONSOLIDATED BALANCE SHEETS
STOCKHOLDERS’ EQUITY
| June 30, | ||||||||
| 2023 | 2022 | |||||||
| Stockholders’ Equity: | ||||||||
| Class A non-voting preferred stock $par value; shares authorized at June 30, 2023 and 2022, issued and outstanding at June 30, 2023 and 2022 | $ | $ | ||||||
| Preferred stock $par value; shares authorized at June 30, 2023 and 2022, issued and outstanding – | ||||||||
| Common stock $par value; shares authorized at June 30, 2023 and 2022, and issued at June 30, 2023 and 2022, respectively and outstanding at June 30, 2023 and 2022, respectively | ||||||||
| Class B convertible common stock (10 votes per share) $par value; shares authorized at June 30, 2023 and 2022, issued and outstanding at June 30, 2023 and 2022 | ||||||||
| Class C common stock (25 votes per share) $par value; shares authorized at June 30, 2023 and 2022, issued and outstanding at June 30, 2023 and 2022 | ||||||||
| Paid-in capital in excess of par value | ||||||||
| Accumulated deficit | ( | ) | ( | ) | ||||
| Treasury stock, at cost – and shares of common stock at June 30, 2023 and 2022, respectively | ( | ) | ( | ) | ||||
| Total Fonar Corporation’s Stockholders’ Equity | ||||||||
| Noncontrolling interests | ( | ) | ( | ) | ||||
| Total Stockholders’ Equity | ||||||||
| Total Liabilities and Stockholders’ Equity | $ | $ | ||||||
See accompanying notes to consolidated financial statements.
| Page 45 |
FONAR CORPORATION AND SUBSIDIARIES
CONSOLIDATED STATEMENTS OF INCOME
| For the Years Ended June 30, | ||||||||
| 2023 | 2022 | |||||||
| Revenues | ||||||||
| Patient fee revenue, net of contractual allowances and discounts | $ | $ | ||||||
| Product sales – net | ||||||||
| Service and repair fees – net | ||||||||
| Service and repair fees – related parties – net | ||||||||
| Management and other fees – net | ||||||||
| Management and other fees – related party medical practices – net | ||||||||
| Total Revenues – Net | ||||||||
| Costs and Expenses | ||||||||
| Costs related to product sales | ||||||||
| Costs related to service and repair fees | ||||||||
| Costs related to service and repair fees – related parties | ||||||||
| Costs related to patient fee revenue | ||||||||
| Costs related to management and other fees | ||||||||
| Costs related to management and other fees – related party medical practices | ||||||||
| Research and development | ||||||||
| Selling, general and administrative expenses | ||||||||
| Total Costs and Expenses | ||||||||
| Income from Operations | ||||||||
| Other Income and (Expenses): | ||||||||
| Interest expense | ( | ) | ( | ) | ||||
| Investment income | ||||||||
| Other (expense) income | ( | ) | ||||||
| Income before provision for income taxes and noncontrolling interests | ||||||||
| Provision for Income Taxes | ( | ) | ( | ) | ||||
| Net Income | $ | $ | ||||||
| Net Income – Noncontrolling Interests | ( | ) | ( | ) | ||||
| Net Income – Attributable to FONAR | $ | $ | ||||||
See accompanying notes to consolidated financial statements.
| Page 46 |
FONAR CORPORATION AND SUBSIDIARIES
CONSOLIDATED STATEMENTS OF INCOME (Continued)
| For the Years Ended June 30, | ||||||||
| 2023 | 2022 | |||||||
| Net Income Available to Common Stockholders | $ | 8,801,974 | $ | 11,690,796 | ||||
| Net Income Available to Class A Non-Voting Preferred Stockholders | $ | 427,666 | $ | 559,072 | ||||
| Net Income Available to Class C Common Stockholders | $ | 146,136 | $ | 191,038 | ||||
| Basic Net Income Per Common Share Available to Common Stockholders | $ | 1.35 | $ | 1.78 | ||||
| Diluted Net Income Per Common Share Available to Common Stockholders | $ | 1.32 | $ | 1.75 | ||||
| Basic and Diluted Income Per Share – Class C Common | $ | 0.38 | $ | 0.50 | ||||
| Weighted Average Basic Shares Outstanding – Common Stockholders | 6,539,376 | 6,554,209 | ||||||
| Weighted Average Diluted Shares Outstanding – Common Stockholders | 6,666,880 | 6,681,713 | ||||||
| Weighted Average Basic and Diluted Shares Outstanding – Class C Common | 382,513 | 382,513 | ||||||
See accompanying notes to consolidated financial statements.
| Page 47 |
FONAR CORPORATION AND SUBSIDIARIES
CONSOLIDATED STATEMENTS OF STOCKHOLDERS’ EQUITY
FOR THE YEARS ENDED JUNE 30, 2023 AND 2022
| Class A Non-Voting Preferred | Common Shares | Stock Amount | Class C Common Stock | |||||||||||||
| Balance, July 1, 2021 | $ | $ | $ | |||||||||||||
| Net income | — | |||||||||||||||
| Stock issued to employees under stock bonus plans | — | |||||||||||||||
| Distributions to noncontrolling interests | — | |||||||||||||||
| Balance, June 30, 2022 | $ | $ | $ | |||||||||||||
| Net income | — | |||||||||||||||
| Purchase of treasury shares | — | |||||||||||||||
| Cancellation of shares | ( | ) | ( | ) | ||||||||||||
| Distributions to noncontrolling interests | — | |||||||||||||||
| Balance, June 30, 2023 | $ | $ | $ | |||||||||||||
| Paid-in Capital in Excess of Par Value | Accumulated Deficit | |||||||
| Balance, July 1, 2021 | $ | $ | ( | ) | ||||
| Net income | ||||||||
| Buyout of noncontrolling interests | ( | ) | ||||||
| Distributions to noncontrolling interests | ||||||||
| Balance, June 30, 2022 | $ | $ | ( | ) | ||||
| Net income | ||||||||
| Purchase of treasury shares | ||||||||
| Cancellation of shares | ( | ) | ||||||
| Distributions to noncontrolling interests | ||||||||
| Balance, June 30, 2023 | $ | $ | ( | ) | ||||
See accompanying notes to consolidated financial statements.
| Page 48 |
FONAR CORPORATION AND SUBSIDIARIES
CONSOLIDATED STATEMENTS OF STOCKHOLDERS’ EQUITY
FOR THE YEARS ENDED JUNE 30, 2023 AND 2022
| Treasury Stock | Noncontrolling Interests | Total | ||||||||||
| Balance, July 1, 2021 | $ | ( | ) | $ | ( | ) | $ | |||||
| Net income | ||||||||||||
| Buyout of noncontrolling interests | ( | ) | ||||||||||
| Distributions to noncontrolling interests | ( | ) | ( | ) | ||||||||
| Balance, June 30, 2022 | $ | ( | ) | $ | ( | ) | $ | |||||
| Net income | ||||||||||||
| Purchase of treasury shares | ( | ) | ( | ) | ||||||||
| Cancellation of shares | ||||||||||||
| Distributions to noncontrolling interests | ( | ) | ( | ) | ||||||||
| Balance, June 30, 2023 | $ | ( | ) | $ | ( | ) | $ | |||||
See accompanying notes to consolidated financial statements.
| Page 49 |
FONAR CORPORATION AND SUBSIDIARIES
CONSOLIDATED STATEMENTS OF CASH FLOWS
| For the Years Ended June 30, | ||||||||
| CASH FLOWS FROM OPERATING ACTIVITIES | 2023 | 2022 | ||||||
| Net Income | $ | $ | ||||||
| Adjustments to reconcile net income to net cash provided by operating activities: | ||||||||
| Depreciation and amortization | ||||||||
| Provision for bad debts | ||||||||
| Deferred income tax - net | ||||||||
| Amortization on right-of-use assets | ||||||||
| Loss on disposition of fixed assets | ||||||||
| Gain on forgiveness of PPP loan | ( | ) | ||||||
| (Increase) decrease in operating assets, net: | ||||||||
| Accounts, medical and management fee receivables | ( | ) | ( | ) | ||||
| Notes receivable | ( | ) | ||||||
| Inventories | ( | ) | ( | ) | ||||
| Prepaid expenses and other current assets | ( | ) | ||||||
| Other assets | ||||||||
| Increase (decrease) in operating liabilities, net: | ||||||||
| Accounts payable | ( | ) | ||||||
| Other current liabilities | ( | ) | ( | ) | ||||
| Customer advances | ( | ) | ||||||
| Operating lease liabilities | ( | ) | ( | ) | ||||
| Financing lease liabilities | ( | ) | ( | ) | ||||
| Contract liabilities | ( | ) | ||||||
| Other liabilities | ( | ) | ( | ) | ||||
| NET CASH PROVIDED BY OPERATING ACTIVITIES | ||||||||
See accompanying notes to consolidated financial statements.
| Page 50 |
FONAR CORPORATION AND SUBSIDIARIES
CONSOLIDATED STATEMENTS OF CASH FLOWS
Continued
| For the Years Ended June 30, | ||||||||
| 2023 | 2022 | |||||||
| CASH FLOWS FROM INVESTING ACTIVITIES | ||||||||
| Purchases of property and equipment | ( | ) | ( | ) | ||||
| Proceeds of Short-term investment | ( | ) | ( | ) | ||||
| Purchase of noncontrolling interests | ( | ) | ||||||
| Cost of patents | ( | ) | ( | ) | ||||
| NET CASH USED IN INVESTING ACTIVITIES | ( | ) | ( | ) | ||||
CASH FLOWS FROM FINANCING ACTIVITIES: | ||||||||
| Repayment of borrowings and capital lease obligations | ( | ) | ( | ) | ||||
| Purchase of treasury stock | ( | ) | ||||||
| Distributions to noncontrolling interests | ( | ) | ( | ) | ||||
| NET CASH USED IN FINANCING ACTIVITIES | ( | ) | ( | ) | ||||
NET INCREASE IN CASH AND CASH EQUIVALENTS | ||||||||
| CASH AND CASH EQUIVALENTS - BEGINNING OF YEAR | ||||||||
| CASH AND CASH EQUIVALENTS - END OF YEAR | $ | $ | ||||||
See accompanying notes to consolidated financial statements.
| Page 51 |
FONAR CORPORATION AND SUBSIDIARIES
NOTES TO CONSOLIDATED FINANCIAL STATEMENTS
JUNE 30, 2023 and 2022
NOTE 1 - DESCRIPTION OF BUSINESS AND LIQUIDITY AND CAPITAL RESOURCES
Description of Business
FONAR Corporation (the “Company” or “FONAR”) is a Delaware corporation, which was incorporated on July 17, 1978. FONAR is engaged in the research, development, production and marketing of medical scanning equipment, which uses principles of Magnetic Resonance Imaging (“MRI”) for the detection and diagnosis of human diseases. In addition to deriving revenues from the direct sale of MRI equipment, revenue is also generated from our installed-base of customers through our service and upgrade programs.
FONAR, through its wholly-owned subsidiary Health Management Corporation of America (“HMCA”) provides comprehensive management services to diagnostic imaging facilities. The services provided by the Company include development, administration, leasing of office space, facilities and medical equipment, provision of supplies, staffing and supervision of non-medical personnel, legal services, accounting, billing and collection and the development and implementation of practice growth and marketing strategies.
On
July 1, 2015, the Company restructured the corporate organization of the management of diagnostic imaging centers segment of our business.
The reorganization was structured to more completely integrate the operations of Health Management Corporation of America and HDM. Imperial
contributed all of its assets (which were utilized in the business of Health Management Corporation of America) to HDM and received a
The global pandemic of COVID-19 has caused turbulence and uncertainty in the United States and international markets and economies which has adversely effected our workforce, liquidity, financial conditions, revenues, profitability and business operations. The Company was able to enact certain decisions to allow the Company to navigate the global pandemic and from further losses, additional decreases in scan volume and avoid any significant disruption of the business. The Company must now take into account the severity, duration and recurrence of new strains of the COVID-19 virus which adds a new dimension to the challenges and uncertainty facing our business and the world economy in general. Although we are unable to predict if there will be additional consequences on our operations from the continuing global pandemic of COVID-19, the Company believes with the positive cash flows, low debt and cash on hand, it will be able to continue operations going forward.
| Page 52 |
FONAR CORPORATION AND SUBSIDIARIES
NOTES TO CONSOLIDATED FINANCIAL STATEMENTS
JUNE 30, 2023 and 2022
NOTE 2 - SUMMARY OF SIGNIFICANT ACCOUNTING POLICIES
Principles of Consolidation
The consolidated financial statements include the accounts of FONAR Corporation, its majority and wholly-owned subsidiaries and partnerships. The operating activities of subsidiaries are included in the accompanying consolidated statements from the date of acquisition. All significant intercompany accounts and transactions have been eliminated in consolidation.
Use of Estimates
The preparation of the consolidated financial statements in conformity with accounting principles generally accepted in the United States requires management to make estimates and assumptions that affect the reported amounts of assets and liabilities and disclosure of contingent assets and liabilities in the consolidated financial statements and accompanying notes. The most significant estimates relate to receivable allowances, intangible assets, income taxes and related tax asset valuation allowances, useful lives of property and equipment, contingencies, revenue recognition and the assessment of litigation. In addition, healthcare industry reforms and reimbursement practices will continue to impact the Company’s operations and the determination of contractual and other allowance estimates. Actual results could differ from those estimates.
Inventories
Inventories consist of purchased parts, components and supplies, as well as work-in-process, and are stated at the lower of cost, determined on the first-in, first-out method, or market.
Property and Equipment
Property
and equipment procured in the normal course of business is stated at cost less accumulated depreciation. Property and equipment purchased in
connection with an acquisition is stated at its estimated fair value, generally based on an appraisal. Property and equipment is
being depreciated for financial accounting purposes using the straight-line method over their estimated useful lives. Leasehold
improvements are being amortized over the shorter of the useful life or the remaining lease term. Upon retirement or other
disposition of these assets, the cost and related accumulated depreciation of these assets are removed from the accounts and the
resulting gains or losses are reflected in the results of operations. Expenses for maintenance and repairs are charged to
operations. Renewals and betterments are capitalized. Maintenance and repair expenses totaled approximately $
| Estimated Useful Life in Years for Property and Equipment | |||
| Diagnostic equipment | |||
| Research, development and demonstration equipment | |||
| Machinery and equipment | |||
| Furniture and fixtures | |||
| Leasehold improvements | |||
| Building |
| Page 53 |
FONAR CORPORATION AND SUBSIDIARIES
NOTES TO CONSOLIDATED FINANCIAL STATEMENTS
JUNE 30, 2023 and 2022
NOTE 2 - SUMMARY OF SIGNIFICANT ACCOUNTING POLICIES (CONTINUED)
Long-Lived Assets
The Company periodically assesses the recoverability of long-lived assets, including property and equipment and intangibles, other than goodwill, when there are indications of potential impairment, based on estimates of undiscounted future cash flows. The amount of impairment is calculated by comparing anticipated discounted future cash flows with the carrying value of the related asset. In performing this analysis, management considers such factors as current results, trends, and future prospects, in addition to other economic factors.
Other Intangible Assets
1) Patents and Copyrights
Amortization
is calculated on the straight-line basis over
2) Non-Competition Agreements
The
non-competition agreements are being amortized on the straight-line basis over the length of the agreement (
3) Customer Relationships
Amortization
is calculated on the straight line basis over
Goodwill
Generally accepted accounting principles in the United States require the Company to perform a goodwill impairment test annually at the end of each fiscal year and more frequently when negative conditions or a triggering event arises. Impairment of goodwill is tested at the reporting unit level by comparing the reporting unit’s carrying amount, including goodwill to the fair value of the reporting unit. If the carrying amount of the reporting unit exceeds its fair value, goodwill is considered potentially impaired and a second step is performed to measure the amount of impairment loss, if any.
Acquired assets and assumed liabilities
Pursuant to ASC No. 805, “Business Combinations”, if the initial accounting for a business combination is incomplete by the end of the reporting period in which the combination occurs, but during the allowed measurement period not to exceed one year from the acquisition date, the Company adjusts the provisional amounts recognized at the acquisition date by means of adjusting the amount recognized for goodwill.
| Page 54 |
FONAR CORPORATION AND SUBSIDIARIES
NOTES TO CONSOLIDATED FINANCIAL STATEMENTS
JUNE 30, 2023 and 2022
NOTE 2 - SUMMARY OF SIGNIFICANT ACCOUNTING POLICIES (CONTINUED)
Revenue Recognition
Revenue on sales contracts for scanners, included in “product sales” in the accompanying consolidated statements of operations, is recognized under the percentage-of-completion method in accordance with FASB ASC 606, “Revenue Recognition – Construction-Type and Production-Type Contracts”. The Company manufactures its scanners under specific contracts that provide for progress payments. Production and installation take approximately three to six months.
Revenue on scanner service contracts is recognized on the straight-line method over the related contract period, usually one year.
Revenue from product sales (upgrades and supplies) is recognized upon shipment.
Revenue
under management contracts is recognized based upon contractual agreements for management services rendered by the Company primarily
under various long-term agreements with various medical providers (the “PCs”). As of June 30, 2023, the Company has
The Company currently recognizes revenue in accordance with the recognition accounting standard issued by the Financial Accounting Standards Board (“FASB”) and codified in the ASC as topic 606 (“ASC 606”). The revenue recognition standard in ASC 606 outlines a single comprehensive model for recognizing revenue as performance obligations, defined in a contract with a customer as goods or services transferred to the customer in exchange for consideration, are satisfied. The standard also requires expanded disclosures regarding the Company’s revenue recognition policies and significant judgments employed in the determination of revenue.
| Page 55 |
.
FONAR CORPORATION AND SUBSIDIARIES
NOTES TO CONSOLIDATED FINANCIAL STATEMENTS
JUNE 30, 2023 and 2022
NOTE 2 - SUMMARY OF SIGNIFICANT ACCOUNTING POLICIES (CONTINUED)
Revenue Recognition (Continued)
Our revenues generally relate to net patient fees received from various payers and patients themselves under contracts in which our performance obligations are to provide diagnostic services to the patients. Revenues are recorded during the period our obligations to provide diagnostic services are satisfied. Our performance obligations for diagnostic services are generally satisfied over a period of less than one day. The contractual relationships with patients, in most cases, also involve a third-party payer (Medicare, Medicaid, managed care health plans and commercial insurance companies, including plans offered through the health insurance exchanges) and the transaction prices for the services provided are dependent upon the terms provided by (Medicare and Medicaid) or negotiated with (managed care health plans and commercial insurance companies) the third-party payers. The payment arrangements with third-party payers for the services we provide to the related patients typically specify payments at amounts less than our standard charges and generally provide for payments based upon predetermined rates per diagnostic services or discounted fee-for-service rates. Management continually reviews the contractual estimation process to consider and incorporate updates to laws and regulations and the frequent changes in managed care contractual terms resulting from contract renegotiations and renewals.
The Company’s patient fee revenues, net of contractual allowances and discounts less the provision for bad debts for the years ended June 30, 2023 and 2022 are summarized in the following table.
| Patient Fee Revenue - Net | ||||||||
| For the Years Ended June 30 | ||||||||
| 2023 | 2022 | |||||||
| Commercial Insurance/ Managed Care | $ | $ | ||||||
| Medicare/Medicaid | ||||||||
| Workers’ Compensation/Personal Injury | ||||||||
| Other | ||||||||
| Net Patient Fee Revenue | $ | $ | ||||||
Research and Development Costs
Research and development costs are charged to expense as incurred. The costs of equipment that are acquired or constructed for research and development activities, and have alternative future uses (either in research and development, marketing or production), are classified as property and equipment and depreciated over their estimated useful lives.
| Page 56 |
FONAR CORPORATION AND SUBSIDIARIES
NOTES TO CONSOLIDATED FINANCIAL STATEMENTS
JUNE 30, 2023 and 2022
NOTE 2 - SUMMARY OF SIGNIFICANT ACCOUNTING POLICIES (CONTINUED)
Advertising Costs
Advertising
costs are expensed as incurred. Advertising expense approximated $
Income Taxes
Deferred tax assets and liabilities are determined based on the difference between the financial statement carrying amounts and tax basis of assets and liabilities using enacted tax rates in effect in the years in which the differences are expected to reverse.
Customer Advances
Cash advances and progress payments received on sales orders are reflected as customer advances until such time as revenue recognition occurs.
Basic earnings per share (“EPS”) is computed by dividing net income available to common stockholders by the weighted average number of shares of common stock outstanding during the period. In accordance with ASC topic 260-10, “Participating Securities and the Two-Class Method”, the Company used the Two-Class method for calculating basic earnings per share and applied the if converted method in calculating diluted earnings per share for the years ended June 30, 2023 and 2022.
Diluted EPS reflects the potential dilution from the exercise or conversion of all dilutive securities into common stock based on the average market price of common shares outstanding during the period. For the years ended June 30, 2023 and 2022, diluted EPS for common shareholders includes shares upon conversion of Class C Common.
| Page 57 |
FONAR CORPORATION AND SUBSIDIARIES
NOTES TO CONSOLIDATED FINANCIAL STATEMENTS
JUNE 30, 2023 and 2022
NOTE 2 - SUMMARY OF SIGNIFICANT ACCOUNTING POLICIES (CONTINUED)
| June 30, 2023 | ||||||||||||
| Basic | Total | Common Stock | Class C Common Stock | |||||||||
| Numerator: | ||||||||||||
| Net income available to common stockholders | $ | $ | $ | |||||||||
| Denominator: | ||||||||||||
| Weighted average shares outstanding | ||||||||||||
| Basic income per common share | $ | $ | $ | |||||||||
| Diluted | ||||||||||||
| Denominator: | ||||||||||||
| Weighted average shares outstanding | ||||||||||||
| Class C Common Stock | ||||||||||||
| Total Denominator for diluted earnings per share | ||||||||||||
| Diluted income per common share | $ | $ | ||||||||||
| June 30, 2022 | ||||||||||||
| Basic | Total | Common Stock | Class C Common Stock | |||||||||
| Numerator: | ||||||||||||
| Net income available to common stockholders | $ | $ | $ | |||||||||
| Denominator: | ||||||||||||
| Weighted average shares outstanding | ||||||||||||
| Basic income per common share | $ | $ | $ | |||||||||
| Diluted | ||||||||||||
| Denominator: | ||||||||||||
| Weighted average shares outstanding | ||||||||||||
| Class C Common Stock | ||||||||||||
| Total Denominator for diluted earnings per share | ||||||||||||
| Diluted income per common share | $ | $ | ||||||||||
Cash and Cash Equivalents
Cash and cash equivalents includes cash on hand, cash in banks, investments in certificates of deposit with original maturities of 90 days or less, and money market funds.
| Page 58 |
FONAR CORPORATION AND SUBSIDIARIES
NOTES TO CONSOLIDATED FINANCIAL STATEMENTS
JUNE 30, 2023 and 2022
NOTE 2 – SUMMARY OF SIGNIFICANT ACCOUNTING POLICIES (CONTINUED)
Short-Term Investments
Short-term investments include certificates of deposit with original maturities of greater than 90 days.
Concentration of Credit Risk
Cash:
The Company maintains its cash and cash equivalents with various financial institutions, which exceed federally insured limits throughout
the year. At June 30, 2023, the Company had cash on deposit of approximately $
Related
Parties: Net revenues from related parties accounted for approximately
See Note 3 regarding the Company’s concentrations in the healthcare industry.
Fair Value of Financial Instruments
The financial statements include various estimated fair value information at June 30, 2023 and 2022, as required by ASC topic 820, “Disclosures about Fair Value of Financial Instruments”. Such information, which pertains to the Company’s financial instruments, is based on the requirements set forth in that Statement and does not purport to represent the aggregate net fair value to the Company.
The standard establishes a three-tier fair value hierarchy, which prioritizes the inputs used in measuring and revaluing fair value. These tiers include, Level 1, defined as observable inputs such as quoted prices in active markets; Level 2, defined as inputs other than quoted prices in active markets that are either directly or indirectly observable; and Level 3, defined as unobservable inputs in which little or no market data exists, therefore requiring an entity to develop its own assumptions.
The following methods and assumptions were used to estimate the fair value of each class of financial instruments for which it is practicable to estimate that value:
Cash and cash equivalents: The carrying amount approximates fair value because of the short-term maturity of those instruments.
Short-term investments: The carrying amount approximates fair value because of the short-term maturity of those instruments. Such amounts include Certificates of Deposits with original maturities greater than 90 days. These securities are classified as Level 1.
| Page 59 |
FONAR CORPORATION AND SUBSIDIARIES
NOTES TO CONSOLIDATED FINANCIAL STATEMENTS
JUNE 30, 2023 and 2022
NOTE 2 - SUMMARY OF SIGNIFICANT ACCOUNTING POLICIES (CONTINUED)
Fair Value of Financial Instruments (Continued)
Receivable and accounts payable: The carrying amounts approximate fair value because of the short maturity of those instruments.
Notes receivable: The carrying amount approximates fair value because the discounted present value of the cash flow generated by the parties approximates the carrying value of the amounts due to the Company.
Long-term debt and notes payable: The carrying amounts of debt and notes payable approximate fair value due to the length of the maturities, the interest rates being tied to market indices and/or due to the interest rates not being significantly different from the current market rates available to the Company.
All of the Company’s financial instruments are held for purposes other than trading.
Recent Accounting Standards
FASB, the Emerging Issues Task Force and the SEC have issued certain other accounting standards, updates, and regulations as of June 30, 2023 that will become effective in subsequent periods; however, management does not believe that any of those updates would have significantly affected our financial accounting measures or disclosures had they been in effect during 2023 or 2022, and it does not believe that any of those standards will have a significant impact on our consolidated financial statements at the time they become effective.
NOTE 3 – ACCOUNTS RECEIVABLE, MEDICAL RECEIVABLE AND MANAGEMENT AND OTHER FEES RECEIVABLE
Accounts Receivable
Credit risk with respect to the Company’s accounts receivable related to product sales and service and repair fees is limited due to the customer advances received prior to the commencement of work performed and the billing of amounts to customers as sub-assemblies are completed. Service and repair fees are billed on a monthly or quarterly basis and the Company does not continue providing these services if accounts receivable become past due. The Company controls credit risk with respect to accounts receivable from service and repair fees through its credit evaluation process, credit limits, monitoring procedures and reasonably short collection terms. The Company performs ongoing credit authorizations before a product sales contract is entered into or service and repair fees are provided.
| Page 60 |
FONAR CORPORATION AND SUBSIDIARIES
NOTES TO CONSOLIDATED FINANCIAL STATEMENTS
JUNE 30, 2023 and 2022
NOTE 3 – ACCOUNTS RECEIVABLE, MEDICAL RECEIVABLE AND MANAGEMENT AND OTHER FEES RECEIVABLE (CONTINUED)
Long Term Accounts Receivable
The Company will generate revenue from long-term, non-cancellable contracts to provide service and repair services. Future revenue to be recognized over the following two years at June 30, 2023 is as follows:
| Receivables - Non Current - Net | ||||
| 2025 | $ | |||
| 2026 | ||||
| Total | $ | |||
Medical Receivable
Medical receivables are due under fee-for-service contracts from third-party payors, such as hospitals, government sponsored healthcare programs, patient’s legal counsel and directly from patients. Substantially all the revenue relates to patients residing in Florida. The carrying amount of the medical receivable is reduced by an allowance that reflects management’s best estimate of the amounts that will not be collected. The Company determines allowances for contractual adjustments and uncollectible accounts based on specific agings, specific payor collection issues that have been identified and based on payor classifications and historical experience at each site.
Management and Other Fees Receivable
The Company’s receivables from the related and non-related professional corporations (“PCs”) substantially consist of fees outstanding under management agreements. Payment of the outstanding fees is dependent on collection by the PCs of fees from third-party medical reimbursement organizations, principally insurance companies and health management organizations.
Payment
of the management fee receivables from the PC’s may be impaired by the inability of the PC’s to collect in a timely manner
their medical fees from the third-party payors, particularly insurance carriers covering automobile no-fault and workers compensation
claims due to longer payment cycles and rigorous informational requirements and certain other disallowed claims. Approximately
| Page 61 |
FONAR CORPORATION AND SUBSIDIARIES
NOTES TO CONSOLIDATED FINANCIAL STATEMENTS
JUNE 30, 2023 and 2022
NOTE 3 – ACCOUNTS RECEIVABLE, MEDICAL RECEIVABLE AND MANAGEMENT AND OTHER FEES RECEIVABLE (CONTINUED)
Net
revenues from management and other fees charged to the related party medical practices accounted for approximately
Tallahassee Magnetic Resonance Imaging, PA, Stand Up MRI of Boca Raton, PA and Stand Up MRI & Diagnostic Center, PA (all related party medical practices) entered into a guaranty agreement, pursuant to which they cross guaranteed all management fees which are payable to the Company, which have arisen under each individual management agreement.
The following table sets forth the number of our facilities for the years ended June 30, 2023 and 2022.
| Total Facilities | ||||||||
| For the Year Ended June 30, | ||||||||
| 2023 | 2022 | |||||||
| Total Facilities Owned or Managed (at Beginning of Year) | ||||||||
| Facilities Added by: | ||||||||
| Internal development | ||||||||
| Managed Facilities Closed | ( | ) | ||||||
| Total Facilities Owned or Managed (at End of Year) | ||||||||
| Page 62 |
FONAR CORPORATION AND SUBSIDIARIES
NOTES TO CONSOLIDATED FINANCIAL STATEMENTS
JUNE 30, 2023 and 2022
NOTE 4 – INVENTORIES
Inventories included in the accompanying consolidated balance sheets consist of:
| Inventories | ||||||||
| As of June 30, | ||||||||
| 2023 | 2022 | |||||||
| Purchased parts, components and supplies | $ | $ | ||||||
| Work-in-process | ||||||||
| Inventories | $ | $ | ||||||
NOTE 5 - PROPERTY AND EQUIPMENT
Property and equipment, at cost, less accumulated depreciation and amortization, at June 30, 2023 and 2022, is comprised of:
| Property and Equipment | ||||||||
| As of June 30, | ||||||||
| 2023 | 2022 | |||||||
| Diagnostic equipment | $ | $ | ||||||
| Research, development and demonstration equipment | ||||||||
| Machinery and equipment | ||||||||
| Furniture and fixtures | ||||||||
| Leasehold improvements | ||||||||
| Building | ||||||||
| Less: Accumulated depreciation and amortization | ||||||||
| $ | $ | |||||||
Depreciation
and amortization of property and equipment for the years ended June 30, 2023 and 2022 was $
| Page 63 |
FONAR CORPORATION AND SUBSIDIARIES
NOTES TO CONSOLIDATED FINANCIAL STATEMENTS
JUNE 30, 2023 and 2022
NOTE 6 – OPERATING & FINANCING LEASES
In July 2019, the Company adopted ASU 2016-02, Leases (Topic 842). This standard requires lessees to apply a dual approach, classifying leases as either finance or operating leases based upon the principle of whether or not the lease is effectively a financed purchase by the lessee. We have elected the optional transition method to apply the standard as of the effective date and therefore, we will not apply the standard to the comparative periods presented in the consolidated financial statements. We have also elected the transition package of these practical expedients permitted within the standard which eliminates the requirements to reassess prior conclusions about lease identification, lease classification and indirect costs.
The Company accounts for its various operating leases in accordance with Accounting Standards Codification (‘ASC’) 842 – Lease, as updated by ASU 2016-02. At the inception of a lease, the Company recognizes right-of-use lease assets and related lease liabilities measured at present value of future lease payments on its balance sheet. Lease expense is recognized on a straight-line basis over the term of the lease. Our most common initial term varies in length from 2 to 10 years. Including renewal options negotiated with the landlord, we have a total span of 2 to 16 years at the facilities we lease. The Company reviewed its contracts with vendors and customers, determining that its right-to-use lease assets consisted of only office space operating leases. In determining the right-to-use lease assets and liabilities, the Company did recognize lease extension options which the Company feels would be reasonably exercised. Our incremental borrowing rate (“IBR”) used to discount the stream of operating lease payments is closely related to the interest rates available to the Company. A reconciliation of operating and financing lease payments undiscounted cash flows to lease liabilities recognized as of June 30, 2023 is as follows:
Reconciliation of operating and financing lease payments Year Ending June 30, | Operating Lease Payments | Financing Lease Payments | |||||||
| 2024 | $ | $ | |||||||
| 2025 | |||||||||
| 2026 | |||||||||
| 2027 | |||||||||
| 2028 | |||||||||
| Thereafter | |||||||||
| Present value discount | ( | ) | ( | ) | |||||
| Total lease liability | $ | $ | |||||||
| Page 64 |
FONAR CORPORATION AND SUBSIDIARIES
NOTES TO CONSOLIDATED FINANCIAL STATEMENTS
JUNE 30, 2023 and 2022
NOTE 6 – OPERATING & FINANCING LEASES (CONTINUED)
| Weighted Average Remaining Lease Term | ||||
| Operating leases - years | ||||
| Finance lease - years | ||||
| Weighted Average Discount Rate | ||||
| Operating leases | % | |||
| Finance lease | % |
The components of lease expense were as follows:
| Components of Lease Expense | ||||||||
| For Year Ended June 30, | ||||||||
| 2023 | 2022 | |||||||
| Operating lease cost | $ | $ | ||||||
Finance lease cost: | ||||||||
| Depreciation of leased equipment | $ | $ | ||||||
| Interest on lease liabilities | ||||||||
| Total finance lease cost | $ | $ | ||||||
Supplemental cash flow information related to leases as follows:
| Supplemental cash flow information related to leases | ||||||||
| For Year Ended June 30, | ||||||||
| Cash paid for amounts included in the measurement of lease liabilities: | 2023 | 2022 | ||||||
| Operating cash flows from operating leases | $ | $ | ||||||
| Financing cash flows from financing leases | $ | $ | ||||||
| Right-of-use and equipment assets obtained in exchange for lease obligations: | ||||||||
| Operating leases | $ | $ | ||||||
| Page 65 |
FONAR CORPORATION AND SUBSIDIARIES
NOTES TO CONSOLIDATED FINANCIAL STATEMENTS
JUNE 30, 2023 and 2022
NOTE 7 - OTHER INTANGIBLE ASSETS
Other intangible assets, net of accumulated amortization, at June 30, 2023 and 2022 are comprised of:
| Other Intangible Assets - Net | ||||||||
| As of June 30, | ||||||||
| 2023 | 2022 | |||||||
| Capitalized software development costs | $ | $ | ||||||
| Patents and copyrights | ||||||||
| Non-competition agreements | ||||||||
| Customer relationships | ||||||||
| Less: Accumulated amortization | ||||||||
| $ | $ | |||||||
The estimated amortization of other intangible assets for the five years ending June 30, 2028 and thereafter is as follows:
Schedule Of Other Intangible Assets For the Years Ending June 30, | Total | Patents and Copyrights | Customer Relationships | |||||||||||
| 2024 | $ | $ | $ | |||||||||||
| 2025 | ||||||||||||||
| 2026 | ||||||||||||||
| 2027 | ||||||||||||||
| 2028 | ||||||||||||||
| Thereafter | ||||||||||||||
| Other intangible assets - net | $ | $ | $ | |||||||||||
| Page 66 |
FONAR CORPORATION AND SUBSIDIARIES
NOTES TO CONSOLIDATED FINANCIAL STATEMENTS
JUNE 30, 2023 and 2022
NOTE 7 - OTHER INTANGIBLE ASSETS (CONTINUED)
The
weighted average amortization period for other intangible assets is
Information related to the above intangible assets for the years ended June 30, 2023 and 2022 is as follows:
| Other Intangible Assets | ||||||||
For the Year Ended June 30, | ||||||||
| 2023 | 2022 | |||||||
| Balance – Beginning of Year | $ | $ | ||||||
| Amounts capitalized | ||||||||
| Software or patents written off | ||||||||
| Amortization | ( | ) | ( | ) | ||||
| Balance – End of Year | $ | $ | ||||||
Amortization
of patents and copyrights for the years ended June 30, 2023 and 2022 amounted to $
Amortization
of non-competition agreements for the years ended June 30, 2023 and 2022 amounted to $
Amortization
of customer relationships for the years ended June 30, 2023 and 2022 amounted to $
NOTE 8 - CAPITAL STOCK
Common Stock
Class B Common Stock
| Page 67 |
FONAR CORPORATION AND SUBSIDIARIES
NOTES TO CONSOLIDATED FINANCIAL STATEMENTS
JUNE 30, 2023 and 2022
NOTE 8 - CAPITAL STOCK (CONTINUED)
Class C Common Stock
Class A Non-Voting Preferred Stock
On
April 3, 1995, the stockholders ratified a proposal consisting of the creation of a new class of Class A non-voting preferred stock with
special dividend rights and the declaration of a stock dividend on the Company’s common stock consisting of
The Class A non-voting preferred stock is entitled to a special dividend equal to 3-1/4% of the first $10 million, 4-1/2% of the next $20 million and 5-1/2% on amounts in excess of $30 million of the amount of any cash awards or settlements received by the Company in connection with the enforcement of five of the Company’s patents in its patent lawsuits, less the revised special dividend payable on the common stock with respect to one of the Company’s patents.
The Class A non-voting preferred stock participates on an equal per share basis with the common stock in any dividends declared and ranks equally with the common stock on distribution rights, liquidation rights and other rights and preferences (other than the voting rights).
Stock Bonus Plans
On April 23, 2010, the Board approved the 2010 Stock Bonus Plan. The plan entitles the Company to reserve shares of common stock. On August 10, 2010, the Company filed Form S-8 to register the shares. As of June 30, 2023, shares of common stock of FONAR were available for future grant under this plan. For the years ended June 30, 2023 and 2022, shares were issued.
| Page 68 |
FONAR CORPORATION AND SUBSIDIARIES
NOTES TO CONSOLIDATED FINANCIAL STATEMENTS
JUNE 30, 2023 and 2022
NOTE 8 - CAPITAL STOCK (CONTINUED)
Treasury Stock
On
September 13, 2022, the Company adopted a stock repurchase plan. The plan has no expiration date and cannot determine the number of shares
which will be repurchased. On September 26, 2022, the Board of Directors has approved up to $
The
Company utilizes the cost method of accounting to value the treasury stock when repurchasing stock. Under this method, the shares are
valued at the price paid and recorded to treasury stock. When the treasury stock is cancelled, the par value of the stock is reduced
and the additional paid in capital is reduced for the remaining value based upon the original stock sale. For the year ended June 30,
2023, the Company purchased shares
at a cost of $
NOTE 9 – CONTROLLING AND NONCONTROLLING INTERESTS
On
February 13, 2013, the Company entered into an agreement with outside investors to acquire a 50.5% controlling interest in a newly formed
limited liability company, Health Diagnostics Management LLC (HDM). According to the February 13, 2013, LLC operating agreement of HDM
there are two classes of members; Class A members and one Class B member. The Class A members have an ownership interest of
On
March 5, 2013, HDM purchased from Health Diagnostics, LLC (“HD”) and certain of its subsidiaries, a business managing twelve
(
| Page 69 |
FONAR CORPORATION AND SUBSIDIARIES
NOTES TO CONSOLIDATED FINANCIAL STATEMENTS
JUNE 30, 2023 and 2022
NOTE 9 – CONTROLLING AND NONCONTROLLING INTERESTS (CONTINUED)
On
January 8, 2015, the Company purchased
The amount of each class of HDM members’ equity as of June 30, 2023 and 2022 is as follows:
| Class A and B Members' Equity | ||||||||||||||||
| June 30, 2023 | June 30, 2022 | |||||||||||||||
| Class A Members | Class B Member | Class A Members | Class B Member | |||||||||||||
| Opening Members' Equity | ($ | ) | $ | ($ | ) | $ | ||||||||||
| Share of Net Income | $ | $ | $ | $ | ||||||||||||
| Buyout of noncontrolling interests | $ | |||||||||||||||
| Distributions | ($ | ) | $ | ( | ) | ($ | ) | ($ | ) | |||||||
| Ending Members' Equity | ($ | ) | $ | ($ | ) | $ | ||||||||||
| Page 70 |
FONAR CORPORATION AND SUBSIDIARIES
NOTES TO CONSOLIDATED FINANCIAL STATEMENTS
JUNE 30, 2023 and 2022
NOTE 10 - LONG-TERM DEBT, NOTES PAYABLE AND CAPITAL LEASES
Long-term debt, notes payable and capital leases consist of the following:
| Long-term Debt, Notes Payable and Capital Leases | ||||||||
| 2023 | 2022 | |||||||
| Note
payable requiring monthly payments of interest at a rate of | $ | $ | ||||||
| The
revolving credit note was extended to November 15, 2023. The Company can borrow up to $ | ||||||||
| Less: Current portion | ||||||||
| $ | $ | |||||||
The maturities of debt over the next four years are as follows:
| Maturities of Long-term Debt | ||||||
| Years Ending June 30, | ||||||
| 2024 | $ | |||||
| 2025 | ||||||
| 2026 | ||||||
| 2027 | ||||||
| $ | ||||||
| Page 71 |
FONAR CORPORATION AND SUBSIDIARIES
NOTES TO CONSOLIDATED FINANCIAL STATEMENTS
JUNE 30, 2023 and 2022
NOTE 11 - INCOME TAXES
In accordance with ASC 740, “Accounting for Income Taxes” prescribes a recognition threshold and a measurement attribute for the financial statement recognition and measurement of tax positions taken or expected to be taken in a corporate tax return. For those benefits to be recognized, a tax position must be more-likely-than-not to be sustained upon examination by taxing authorities. Differences between tax positions taken or expected to be taken in a tax return and the benefit recognized and measured pursuant to the interpretation are referred to as unrecognized benefits. A liability is recognized (or amount of net operating loss carryforward or amount of tax refundable is reduced) for an unrecognized tax benefit because it represents an enterprise’s potential future obligation to the taxing authority for a tax position that was not recognized as a result of applying the provisions of ASC 740. The Company believes there are no uncertain tax positions in prior year’s tax filings and therefore it has not recorded a liability for unrecognized tax benefits.
In accordance with ASC 740, interest costs related to unrecognized tax benefits are required to be calculated (if applicable) and would be classified as “Interest expense, net. Penalties if incurred would be recognized as a component of “Selling, general and administrative” expenses.
The Company files corporate income tax returns in the United States (federal) and in various state and local jurisdictions. In most instances, the Company is no longer subject to federal, state and local income tax examinations by tax authorities for years prior to 2019.
The
Company has recorded a deferred tax asset of $
Future ownership changes as determined under Section 382 of the Internal Revenue code could further limit the utilization of net operating loss carryforwards. As of June 30, 2023, no such changes in ownership have occurred.
The Inflation Reduction Act (“IRA”) was enacted on August 16, 2022. The IRA includes provisions imposing a 1% excise tax on share repurchases that occur after December 31, 2022 and introduces a 15% corporate alternative minimum tax (“CAMT”) on adjusted financial statement income. The CAMT will be effective for tax years beginning after December 31, 2022. Currently, the Company does not expect the IRA to have a material impact to the Company’s financial statements.
| Page 72 |
FONAR CORPORATION AND SUBSIDIARIES
NOTES TO CONSOLIDATED FINANCIAL STATEMENTS
JUNE 30, 2023 and 2022
NOTE 11 - INCOME TAXES (CONTINUED)
The ultimate realization of deferred tax assets is dependent on the generation of future taxable income during the periods in which temporary differences become deductible or when such net operating losses can be utilized. The Company considers projected future taxable income, the regulatory environment of the industry, and tax planning strategies in making this assessment. At present, the Company believes that it is more likely than not that the benefits from certain deferred tax asset carryforwards, will not all be fully realized. In recognition of this inherent risk, a valuation allowance was established for the partial value of the deferred tax asset, which principally related to certain state net operating losses. A valuation allowance will be maintained until sufficient positive evidence exists to support the reversal of the remainder of the valuation.
The
valuation allowance for deferred tax assets decreased during the year ended June 30, 2023, by approximately $
Components of the provision (benefit) for income taxes are as follows:
| Years Ended June 30, | ||||||||
| Current: | 2023 | 2022 | ||||||
| Federal | $ | $ | ||||||
| State | ||||||||
| Subtotal | ||||||||
| Deferred: | ||||||||
| Federal deferred taxes | ||||||||
| State deferred taxes | ||||||||
| Subtotal | ||||||||
| Provision (Benefit) for Income Taxes - Net | $ | $ | ||||||
| Page 73 |
FONAR CORPORATION AND SUBSIDIARIES
NOTES TO CONSOLIDATED FINANCIAL STATEMENTS
JUNE 30, 2023 and 2022
NOTE 11 - INCOME TAXES (CONTINUED)
A reconciliation of the federal statutory income tax rate to the Company’s effective tax rate as reported is as follows:
| Reconciliation Of Federal Statutory Income Tax Rate To Company’s Effective Tax Rate | ||||||||
| Years Ended June 30, | ||||||||
| 2023 | 2022 | |||||||
| Taxes at federal statutory rate | % | % | ||||||
| State and local income taxes (benefit), net of federal benefit | % | % | ||||||
| Noncontrolling interest | ( | )% | ( | )% | ||||
| Expiration of tax credits | % | % | ||||||
| Return to provision adjustments | ( | )% | % | |||||
| New York state audit settlement | % | |||||||
| Change in the valuation allowance | ( | )% | ( | )% | ||||
| Other | % | ( | )% | |||||
| Effective income tax rate | % | % | ||||||
As of June 30, 2023, the Company has net operating loss (“NOL”) carryforwards of approximately $9,110,000 that will be available to offset future taxable income. The utilization of certain of the NOLs is limited by separate return limitation year rules pursuant to Section 1502 of the Internal Revenue Code.
The
Company has, for federal income tax purposes, research and development tax credits and investments tax credits carryforwards aggregating
$
The
Company was also under audit with New York State for income tax and was assessed additional taxes of $
| Page 74 |
FONAR CORPORATION AND SUBSIDIARIES
NOTES TO CONSOLIDATED FINANCIAL STATEMENTS
JUNE 30, 2023 and 2022
NOTE 11 - INCOME TAXES (CONTINUED)
Significant components of the Company’s deferred tax assets and liabilities at June 30, 2023 and 2022 are as follows:
| Components Of Company’s Deferred Tax Assets and Liabilities | ||||||||
| June 30, | ||||||||
| 2023 | 2022 | |||||||
| Deferred tax assets: | ||||||||
| Allowance for doubtful accounts | $ | $ | ||||||
| Non-deductible accruals | ||||||||
| Net operating carryforwards | ||||||||
| Tax credits | ||||||||
| Capitalized research and development | ||||||||
Right of use assets and lease liabilities | ||||||||
| Inventories | ||||||||
| Property and equipment and depreciation | ||||||||
| Deferred Tax Assets - gross | ||||||||
| Valuation allowance | ( | ) | ( | ) | ||||
| Total deferred tax assets | ||||||||
| Property and equipment and depreciation | ( | ) | ||||||
| Intangibles | ( | ) | ( | ) | ||||
| Total deferred tax liabilities | ( | ) | ( | ) | ||||
| Net deferred tax asset | $ | $ | ||||||
NOTE 12 - OTHER CURRENT LIABILITIES
Included in other current liabilities are the following:
| June 30, | ||||||||
| 2023 | 2022 | |||||||
| Accrued salaries, commissions and payroll taxes | $ | $ | ||||||
| Sales tax payable | ||||||||
| State income taxes payable | ||||||||
| Legal and other professional fees | ||||||||
| Accounting fees | ||||||||
| Self-funded health insurance reserve | ||||||||
| Accrued interest and penalty | ||||||||
| Other | ||||||||
| Other current liabilities | $ | $ | ||||||
| Page 75 |
FONAR CORPORATION AND SUBSIDIARIES
NOTES TO CONSOLIDATED FINANCIAL STATEMENTS
JUNE 30, 2023 and 2022
NOTE 13 - COMMITMENTS AND CONTINGENCIES
Leases
The Company rents its operating facilities and certain equipment, pursuant to operating lease agreements expiring at various dates through March 2030. The leases for certain facilities contain escalation clauses relating to increases in real property taxes as well as certain maintenance costs.
Rent
expense for operating leases approximated $
The
Company received approval from the Suffolk County Industrial Development Agency on February 29, 2016 of a
Employee Benefit Plans
The
Company has a non-contributory 401(k) Plan (the “401(k) Plan”). The 401(k) Plan covers all non-union employees who are at
least 21 years of age with no minimum service requirements. There were $
The stockholders of the Company approved the 2000 Employee Stock Purchase Plan (“ESPP”) at the Company’s annual stockholders’ meeting in April 2000. The ESPP provides for eligible employees to acquire common stock of the Company at a discount, not to exceed 15%. This plan has not been put into effect as of June 30, 2023.
Other Matters
The Company is subject to other legal proceedings and claims arising from the ordinary course of its business, including personal injury, customer contract and employment claims besides the claim above. In the opinion of management, and with consultation with legal counsel, the aggregate liability, if any, with respect to such actions, will not have a material adverse effect on the consolidated financial position or results of operations of the Company.
The
Company maintains a self-funded health insurance program with a stop-loss umbrella policy with a third-party insurer to limit the maximum
potential liability for individual claims to $
| Page 76 |
FONAR CORPORATION AND SUBSIDIARIES
NOTES TO CONSOLIDATED FINANCIAL STATEMENTS
JUNE 30, 2023 and 2022
NOTE 13 - COMMITMENTS AND CONTINGENCIES (CONTINUED)
Other Matters (Continued)
The Company regularly analyzes its reserves for incurred but not reported claims, and for reported but not paid claims related to its reinsurance and self-funded insurance programs. The Company believes its reserves are adequate. However, significant judgment is involved in assessing these reserves such as assessing historical paid claims, average lags between the claims’ incurred date, reported dates and paid dates, and the frequency and severity of claims. There may be differences between actual settlement amounts and recorded reserves and any resulting adjustments are included in expense once a probable amount is known. There were no significant adjustments recorded in the years covered by this report.
NOTE 14 - SUPPLEMENTAL CASH FLOW INFORMATION
During
the years ended June 30, 2023 and 2022 the Company paid $
During
the years ended June 30, 2023 and 2022 the Company paid $
During
the years ended June 30, 2023 and 2022, the Company resolved certain sales tax liabilities and was able to reverse accrued interest and
penalties in the amount of $
NOTE 15 – RELATED PARTY TRANSACTIONS
| Page 77 |
FONAR CORPORATION AND SUBSIDIARIES
NOTES TO CONSOLIDATED FINANCIAL STATEMENTS
JUNE 30, 2023 and 2022
NOTE 16 - SEGMENT AND RELATED INFORMATION
The Company provides segment data in accordance with the provisions of ASC 280, “Disclosures about Segments of an Enterprise and Related Information”.
The Company operates in two industry segments - manufacturing and the servicing of medical equipment and management of diagnostic imaging centers.
The accounting policies of the segments are the same as those described in the summary of significant accounting policies. All intersegment sales are market-based. The Company evaluates performance based on income or loss from operations.
Summarized financial information concerning the Company’s reportable segments is shown in the following table:
Summarized Segment Financial Information
| Manufacturing and Servicing of Medical | Management of Diagnostic Imaging | |||||||||||
| Fiscal 2023: | Equipment | Center | Totals | |||||||||
| Net revenues from external customers | $ | $ | $ | |||||||||
| Intersegment net revenues * | $ | $ | $ | |||||||||
| (Loss) Income from operations | $ | ( | ) | $ | $ | |||||||
| Depreciation and amortization | $ | $ | $ | |||||||||
| Total identifiable assets | $ | $ | $ | |||||||||
| Capital expenditures | $ | $ | $ | |||||||||
| Fiscal 2022: | ||||||||||||
| Net revenues from external customers | $ | $ | $ | |||||||||
| Intersegment net revenues * | $ | $ | $ | |||||||||
| (Loss) Income from operations | $ | ( | ) | $ | $ | |||||||
| Depreciation and amortization | $ | $ | $ | |||||||||
| Total identifiable assets | $ | $ | $ | |||||||||
| Capital expenditures | $ | $ | $ | |||||||||
| * |
| Page 78 |
FONAR CORPORATION AND SUBSIDIARIES
NOTES TO CONSOLIDATED FINANCIAL STATEMENTS
JUNE 30, 2023 and 2022
NOTE 16 - SEGMENT AND RELATED INFORMATION (CONTINUED)
Export Product Sales
The
Company’s areas of operations are principally in the United States. The Company had export sales of medical equipment amounting
to
The foreign product sales, as a percentage of product sales to unrelated parties, were made to customers in the following countries:
| For the Years Ended June 30, | ||||||||
| 2023 | 2022 | |||||||
| Dominican Republic | % | |||||||
| Canada | % | % | ||||||
| Germany | % | |||||||
| United Arab Emirates | % | |||||||
| Puerto Rico | % | |||||||
| % | % | |||||||
Foreign Service and Repair Fees
The
Company’s areas of service and repair are principally in the United States. The Company had foreign revenues of service and repair
of medical equipment amounting to
Foreign Service Repair Fees
For the Years Ended June 30, | |||||
| 2023 | 2022 | ||||
| Puerto Rico | % | % | |||
| Switzerland | |||||
| Germany | |||||
| England | |||||
| United Arab Emirates | — | ||||
| Dominican Republic | — | ||||
| Canada | — | ||||
| Greece | |||||
| Australia | |||||
| % | % | ||||
The Company does not have any material assets outside of the United States.
| Page 79 |
FONAR CORPORATION AND SUBSIDIARIES
NOTES TO CONSOLIDATED FINANCIAL STATEMENTS
JUNE 30, 2023 and 2022
NOTE 17 – ALLOWANCE FOR DOUBTFUL ACCOUNTS
The following represents a summary of allowance for doubtful accounts for the years ended June 30, 2023 and 2022 respectively:
Summary of Allowance For Doubtful Accounts
| Description | Balance
June 30, 2022 | Additions (1) | Deductions | Balance
June 30, 2023 | ||||||||||||
| Accounts receivable | $ | $ | $ | $ | ||||||||||||
| Management and other fees receivable | ||||||||||||||||
| Management and other fees receivable - related medical practices | ||||||||||||||||
| Notes receivable | ||||||||||||||||
| Balance | Balance | |||||||||||||||
| Description | June 30, 2021 | Additions | Deductions | June 30, 2022 | ||||||||||||
| Accounts receivable | $ | $ | $ | $ | ||||||||||||
| Management and other fees receivable | ||||||||||||||||
| Management and other fees receivable - related medical practices | ||||||||||||||||
| Notes receivable | ||||||||||||||||
| (1) |
NOTE 18 – SUBSEQUENT EVENTS
The Company evaluates events that have occurred after the balance sheet date, but before the consolidated financial statements are issued.
On
July 31, 2023,
During
August 2023 the Company amended their revolving credit agreement. The agreement was extended to November 15, 2023. The interest
rate on borrowings remains at
As
of August 31, 2023, the Company repurchased shares
at a cost of $
| Page 80 |
FONAR CORPORATION AND SUBSIDIARIES
ITEM 9. CHANGES IN AND DISAGREEMENTS WITH ACCOUNTANTS ON ACCOUNTING AND FINANCIAL DISCLOSURE.
There have been no disagreements with our independent registered public accounting firm or other matters requiring disclosure under Regulation S-K, Item 304(b).
ITEM 9A. CONTROLS AND PROCEDURES
Evaluation of Disclosure Controls and Procedures
As of the end of the period covered by this Annual Report on Form 10-K, we performed an evaluation under the supervision of and with the participation of management, including our Principal Executive Officer and our Acting Principal Financial Officer, of the design and effectiveness of our disclosure controls and procedures (as defined in Rules 13a-15(e) or 15d-15(e) under the Securities Exchange Act of 1934 as amended (the “Exchange Act”). Based upon that evaluation, our Principal Executive Officer and Acting Principal Financial Officer concluded, as of the end of the period covered by this Annual Report that our disclosure controls and procedures were effective.
Management’s Report on Internal Control Over Financial Reporting
Our management is responsible for establishing and maintaining adequate internal control over financial reporting, as is defined in the Exchange Act. Internal control over financial reporting is a process designed to provide reasonable assurance regarding the reliability of our financial reporting and the preparation of financial statements for external reporting purposes in accordance with GAAP.
Because of its inherent limitations, internal control over financial reporting may not prevent or detect misstatements. Also, projections of any evaluation of effectiveness to future periods are subject to the risk that controls may become inadequate because of changes in conditions, or that the degree of compliance with the policies or procedures may deteriorate.
Our management conducted an evaluation of the effectiveness of our internal control over financial reporting based on the framework in Internal Control-Integrated Framework issued by the Committee of Sponsoring Organizations of the Treadway Commission (COSO-2013). Based on this evaluation, our management concluded that our internal control over financial reporting was effective at June 30, 2023.
Based on the COSO criteria, management concluded that our internal controls were effective to prevent material misstatements of the Company’s annual or interim financial statements for the fiscal year ending June 30, 2023.
Changes in Internal Controls over Financial Reporting
There have been no changes in our internal control over financial reporting (as defined in Rule 13a-15(f) under the Exchange Act) during the most recent fiscal quarter and year ended June 30, 2023 that has materially affected, or is reasonably likely to materially affect, our internal control over financial reporting.
| Page 81 |
FONAR CORPORATION AND SUBSIDIARIES
Item 9B. OTHER INFORMATION
None.
Item 9C. DISCLOSURE REGARDING FOREIGN JURISDICTIONS THAT PREVENT INSPECTIONS
None.
PART III
ITEM 10. DIRECTORS AND EXECUTIVE OFFICERS.
Directors serve from the date of their election until the next annual meeting of stockholders and until their successors are elected and qualify. During fiscal 2022, with the exception of Dr. Raymond V. Damadian, who did not receive any fees for serving as a director, each director receives a base fee of $20,000 per annum for his or her service as a director, with greater amounts for additional services on the Board of Directors. Officers serve at the discretion of the Board of Directors.
A majority of our board of directors is composed of independent directors: consisting of, Ronald G. Lehman, Richard E. Turk and Jessica Maher. The outside directors also serve as the members of the audit committee, which is a standing committee of the board of directors having a charter describing its responsibilities.
We have adopted a code of ethics applicable to, among other personnel, our principal executive officer, principal financial officer, controllers and persons performing similar functions. The code is designed to deter wrongdoing and to promote: 1. honest and ethical conduct, including the ethical handling of actual or apparent conflicts of interest between personal and professional relationships; 2. full, fair, accurate, timely and understandable disclosure in reports and documents that we file or submit to the Securities and Exchange Commission and in other public communications we make; 3. compliance with applicable governmental laws, rules and regulations; 4. the prompt internal reporting of violations of the code to an appropriate person or persons identified in the code and 5. accountability for adherence to the code. We will provide a copy of the code to any person who requests a copy. A person may request a copy by writing to FONAR Corporation, 110 Marcus Drive, Melville, New York 11747, to the attention of the Legal Department or Investor Relations.
| Page 82 |
FONAR CORPORATION AND SUBSIDIARIES
The officers and directors of the Company are set forth below:
| Timothy R. Damadian | 59 | Chairman of the Board, President, Chief Executive Officer and Treasurer |
| Luciano B. Bonanni | 68 | Executive Vice President, Chief Operating Officer and acting Principal Financial Officer |
Claudette J.V. Chan |
85 | Director |
Ronald J. Lehman |
47 | Director |
| Richard E. Turk | 39 | Director |
| Jessica Maher | 26 | Director |
Timothy Damadian has been the Chairman of the Board and Treasurer of FONAR since September 7, 2022 and the President and Chief Executive Officer of FONAR since February 11, 2016. From 2010 to 2016 he served as an independent consultant, with a focus on the Company’s MRI facility management business. Timothy Damadian began his career at FONAR in 1985, installing MRI scanners and components for FONAR customers. Over the course of the following 16 years, he held positions of increasing authority, eventually becoming Vice President of Operations. In 1997, Timothy Damadian was appointed President of the newly formed Health Management Corporation of America (HMCA), a wholly-owned subsidiary of FONAR that was formed to manage medical and diagnostic imaging offices. In 2001, Timothy Damadian left FONAR to form Integrity Healthcare Management, Inc., a diagnostic imaging management company that would eventually manage MRI scanning centers in New York and Florida. The company was a success and was sold to Health Diagnostics, LLC in 2007. Mr. Damadian returned to FONAR as a consultant in 2010. He also serves as a Manager of Health Diagnostics Management, LLC, which are subsidiaries of HMCA.
Luciano B. Bonanni has served as Chief Operating Officer (COO) and Executive Vice President (EVP) for FONAR Corporation since June 27, 2016. In September 2022, he was appointed to fill the position of acting Principal Financial Officer. Prior to his appointment as COO, Mr. Bonanni had served the Company as Vice President since 1989, during which time he oversaw general operations, research and development, manufacturing, service, sales, finance, accounting and regulatory compliance. Prior to 1989, Mr. Bonanni held the title of Vice President of Production and Engineering from the time of FONAR’s initial public offering in 1981. Mr. Bonanni joined the Company as an electrical engineer in 1978. He holds a Bachelor of Electrical Engineering degree from Manhattan College.
Claudette J.V. Chan has been a Director of FONAR since October 1987 and Secretary of FONAR since January 2008. Mrs. Chan was employed from 1992 through 1997 by Raymond V. Damadian, M.D. MR Scanning Centers Management Company and since 1997 by HMCA, as “site inspector,” in which capacity she is responsible for supervising and implementing standard procedures and policies for MRI scanning centers. From 1989 to 1994 Mrs. Chan was employed by St. Matthew’s and St. Timothy’s Neighborhood Center, Inc., as the director of volunteers in the “Meals on Wheels” program, a program which cares for the elderly.
| Page 83 |
FONAR CORPORATION AND SUBSIDIARIES
From approximately 1983 to 1989, Mrs. Chan was President of the Claudette Penot Collection, a retail mail-order business specializing in women’s apparel and gifts. Mrs. Chan practiced and taught in the field of nursing until 1973, when her son was born. She received a Bachelor of Science degree in nursing from Cornell University in 1960.
Ronald G. Lehman has been a Director of FONAR since April, 2012, when he was unanimously appointed by the remaining four Directors to fill the vacancy resulting from the death of former Director Robert Djerejian. From October, 2009 to the present, Mr. Lehman has served as Managing Director of Investment Banking with Bruderman Brothers, LLC, a private New York-based broker-dealer registered with the Securities and Exchange Commission and which is a member of the Financial Industry Regulatory Authority (FINRA) and the Securities Investor Protection Corporation (SIPC). Mr. Lehman directly manages all facets of the firm’s transaction processes, from deal origination, to sourcing capital, to negotiating deal structures, through documentation and closing. The firm provides buy and sell-side advisory, capital raising, and consulting services to lower middle-market companies. Mr. Lehman specializes in advising healthcare services companies and has recently completed several recapitalizations in the industry. He also participates in the firm’s merchant banking investments and oversees many of these assignments. From May, 2008 to October, 2009, Mr. Lehman served as Senior Vice President of Acquisitions at Health Diagnostics, LLC, where he managed the company’s acquisition and corporate finance activities. From March, 2000 to May, 2008, Mr. Lehman worked for various Bruderman entities as a buy and sell-side advisor and as a principal in several private equity transactions. From September, 1998 to March, 2000, Mr. Lehman worked at Deutsche Bank Securities, Inc. and last held the position of Associate in their Global Custody Group. Mr. Lehman graduated from Columbia University with a B.A. in 1998.
Richard E. Turk has been a Director of FONAR since June, 2020, when he was appointed to fill the vacancies on the Board of Directors and Audit Committee of the Board of Directors resulting from the death of his predecessor, Robert J. Janoff. Mr. Turk is the Chief Financial Officer of PRISM Vision Group, a private equity-backed, multi-location, outpatient comprehensive eye care practice headquartered in Union, New Jersey. Mr. Turk joined PRISM in November, 2018 as the Chief Development Officer and became CFO in March 2021. Mr. Turk has helped PRISM expand from a single-specialty (retina) provider with 17 locations and 21 physicians to a comprehensive, vertically-integrated, multi-specialty, eye care organization with approximately 190 physicians and more than 90 locations across New Jersey, Pennsylvania, Delaware, Virginia, Washington, DC, and Maryland. Prior to his tenure at PRISM, Mr. Turk was employed by Professional Physical Therapy, a private equity-backed outpatient physical and occupational therapy company headquartered in Uniondale, New Jersey with more than 180 locations across New York, New Jersey, Connecticut, Massachusetts and New Hampshire. During his four years at Professional Physical Therapy, Mr. Turk sourced, analyzed, and completed 32 acquisitions comprised of 116 clinics, expanding the company’s services and adding three states. From 2007 to 2014, Mr. Turk was employed by Bruderman Brothers, a broker dealer involved in investment banking, merchant banking, investment advisory, and consulting for lower middle market companies ($10M-$250M of enterprise value) in a variety of industries, including healthcare. Mr. Turk was Vice President of Bruderman Brothers from 2011 to 2014. Mr. Turk graduated from Columbia University in 2007.
| Page 84 |
FONAR CORPORATION AND SUBSIDIARIES
Jessica Maher has been a Director of FONAR since March 2023, when she was appointed to fill the vacancies on the Board of Directors and Audit Committee resulting from the resignation of her predecessor, John Collins. Mrs. Maher is a staff accountant at Ives & Sultan, LLP in Woodbury, New York, where she is responsible for preparing audited financial statements for various clients, overseeing audit testing areas, audits of 401(k) plans, and personal and company tax returns. Mrs. Maher holds a Bachelor of Science in Accounting with a minor in Accounting Information Systems, and a Master of Science in Accounting from Fairfield University in Fairfield, Connecticut. During her early undergraduate years, Mrs. Maher worked for Tritech Healthcare Management in Melville, New York, where she reviewed patient files, insurance, charts and documents to ensure that the services provided by clients were being properly billed. In her senior year, Mrs. Maher interned at Northwell Health in Westbury, New York, where she supported the financial reporting team for two hospitals, reported into accounts receivable software, and analyzed patient billing records to identify overpayments. Mrs. Maher’s first position out of college was with PriceWaterhouseCoopers in Melville, New York, where she was assigned to two private equity clients, was responsible for a variety of the audit areas, and assisted managers in reviewing financial statements, footnote disclosures, and audit opinions.
| Board Diversity Matrix as of September 12, 2023 | ||
| Total Number of Directors | 5 | |
| Part I: Gender Identity | Female | Male |
| Directors | 2 | 3 |
| Part II: Demographic Background | ||
| White | 2 | 3 |
| Board Diversity Matrix as of September 15, 2022 | |||
| Total Number of Directors | 5 | ||
| Part I: Gender Identity | Female | Male | Did not disclose gender |
| Directors | 1 | 3 | 1 |
| Part II: Demographic Background | |||
| White | 1 | 3 | |
| Did Not Disclose Demographic Background 1 | |||
| Director with Disabilities | 1 | ||
| Page 85 |
FONAR CORPORATION AND SUBSIDIARIES
ITEM 11. EXECUTIVE COMPENSATION.
With the exception of the Chief Executive Officer and the Chairman of the Board of Directors, the compensation of the Company’s executive officers is based on a combination of salary and bonuses based on performance. The Chief Executive Officer and the Chairman of the Board have no understandings with the Company with respect to bonuses, options or other incentives; they are not subject to our general policy later discussed.
The Board of Directors does not have a compensation Committee. The Chief Executive Officer and the Chief Operating Officer participate in the determination of compensation for the Company’s management and other employees.
The Board of Directors has established an audit committee. The members of the committee are Ronald G. Lehman, Richard E. Turk and Jessica Maher.
Our compensation policy includes a combination of salary, commissions, bonuses, stock bonuses and stock options, designed to incentivize our employees. There is no universal plan applicable to all of our employees. The fixed and variable components of our employees’ compensation tend to be individualized, based on a combination of the employees’ performance, responsibilities and position, our assessment of how best to motivate a person in such a position and the needs and preferences of the particular employees, as negotiated between employees and their supervisors or management.
There is set forth in the following Summary Compensation Table the compensation provided by us during fiscal 2023, 2022 and 2021 to our Principal Executive Officer, and our acting Principal Financial Officer. There is set forth in the following Outstanding Equity Awards Table and Director Compensation Table the required information.
| I. | SUMMARY COMPENSATION TABLE |
(Reflects information up to end of Fiscal 2023)
| Name and All Other Principal Position | Year | Salary ($) | Cash
Bonuses ($) | Stock
Awards ($) | Total Compensation ($) | |||||||||||||||
| (a) | (b) | (c) | (d) | (e) | (f) | |||||||||||||||
| Timothy R. Damadian | 2023 | $ | 0 | $ | 152,900 | $ | 0 | $ | 152,900 | |||||||||||
| President, Principal | 2022 | $ | 0 | $ | 305,800 | $ | 0 | $ | 305,800 | |||||||||||
| Executive Officer | 2021 | $ | 0 | $ | 155,800 | $ | 0 | $ | 155,800 | |||||||||||
| Raymond V. Damadian | 2023 | $ | 23,553 | $ | 305,800 | $ | 0 | $ | 329,353 | |||||||||||
| Chairman of the Board, | 2022 | $ | 153,095 | $ | 305,800 | $ | 0 | $ | 458,895 | |||||||||||
| Treasurer and | 2021 | $ | 153,095 | $ | 305,800 | $ | 0 | $ | 458,895 | |||||||||||
| Principal Financial Officer | ||||||||||||||||||||
| Luciano Bonanni | 2023 | $ | 143,416 | $ | 305,800 | $ | 0 | $ | 449,216 | |||||||||||
| Chief Operating Officer and | 2022 | $ | 148,572 | $ | 305,800 | $ | 0 | $ | 454,372 | |||||||||||
| Executive Vice President | 2021 | $ | 146,496 | $ | 0 | $ | 152,931 | $ | 298,969 | |||||||||||
| Page 86 |
FONAR CORPORATION AND SUBSIDIARIES
| II. | OUTSTANDING EQUITY AWARDS AT FISCAL YEAR-END |
| Name | Number Of Securities Underlying Unexercised Options (#) Exercisable | Option Exercise Price ($) | Option Exercise Expiration Date | |||||||
| (a) | (b) | (c) | ||||||||
| Raymond V. Damadian, Chairman of the Board, Treasurer and Principal Financial Officer | 0 | 0 | N/A | |||||||
| Timothy R. Damadian, President and Principal Executive Officer | 0 | 0 | N/A | |||||||
| Luciano Bonanni, Chief Operating Officer, Executive Vice President and acting Principal Financial Officer | 0 | 0 | N/A | |||||||
| III | DIRECTOR COMPENSATION |
The following table shows the compensation paid to the Directors for fiscal 2023:
| Name | Fees
earned in pad in cash ($) | Stock awards ($) | Option awards ($) | Non-equity
incentive plan compen- sation | Nonqualified
deferred compen- sation earnings ($) | All
other compen- sation ($) | Total
($) | |||||||||||||||||||||
| (a) | (b) | (c) | (d) | (e) | (f) | (g) | (h) | |||||||||||||||||||||
| A. Claudette J.V. Chan | $ | 20,000 | 0 | 0 | 0 | 0 | 38,880 | $ | 58,880 | |||||||||||||||||||
| B. Ronald G. Lehman | $ | 20,000 | 0 | 0 | 0 | 0 | 60,000 | $ | 80,000 | |||||||||||||||||||
| C. Richard E. Turk | $ | 20,000 | 0 | 0 | 0 | 0 | $ | 15,000 | $ | 35,000 | ||||||||||||||||||
| D. Jessica Maher | $ | 9,423 | 0 | 0 | 0 | 0 | $ | 0 | $ | 9,423 | ||||||||||||||||||
| E. John Collins | $ | 15,384 | 0 | 0 | 0 | 0 | $ | 44,038 | $ | 59,423 | ||||||||||||||||||
EMPLOYEE COMPENSATION PLANS
FONAR adopted its 2010 Stock Bonus Plan, on June 28, 2010. This Plan permits FONAR to issue an aggregate of 2,000,000 shares of common stock of FONAR as bonus or compensation. As of June 30, 2023, 450,177 shares were available for issuance.
| Page 87 |
FONAR CORPORATION AND SUBSIDIARIES
ITEM 12. SECURITY OWNERSHIP OF CERTAIN BENEFICIAL OWNERS AND MANAGEMENT.
The following table sets forth the number and percentage of shares of FONAR’s securities held by each director, by each person known by us to own in excess of five percent of FONAR’s voting securities and by all officers and directors as a group as of September 1, 2023.
| Name and Address of Beneficial Owner (1) | Shares Beneficially Owned | Percent of Class |
||||
| Timothy R. Damadian, as trustee of the Fonar Class C Trust | ||||||
| c/o FONAR Corporation, Melville, New York | ||||||
| Class C Stock | 382,447 | 99.98 | % | |||
| Kayne Anderson Rudnick | ||||||
| Investment Management LLC | ||||||
| 1800 Avenue of the Stars, 2nd Floor | ||||||
| Los Angeles, CA 90067 | ||||||
| Common Stock | 628,700 | 9.75 | % | |||
| Dimensional Fund Advisors LP | ||||||
| Building One | ||||||
| 6300 Bee Cave Road | ||||||
| Austin, Texas 78746 | ||||||
| Common Stock | 409,855 | 6.35 | % | |||
| The Vanguard Group, Inc. | ||||||
| 100 Vanguard Boulevard | ||||||
| Malvern, PA 19355-2331 | ||||||
| Common Stock | 373,859 | 5.80 | % | |||
| Timothy R. Damadian, | ||||||
| Chairman of the Board, President | ||||||
| Chief Executive Officer and Treasurer | ||||||
| Common Stock | 42,700 | * | ||||
| Class A Preferred | 800 | * | ||||
| Page 88 |
FONAR CORPORATION AND SUBSIDIARIES
Continued:
| Name and Address of Beneficial Owner (1) | Shares Beneficially Owned | Percent of Class | ||||
| Luciano B. Bonanni, | ||||||
| Executive Vice President, | ||||||
| Chief
Operating Officer and acting Principal Financial Officer | ||||||
| Common Stock | 54,253 | * | ||||
| Class A Preferred | 1,285 | * | ||||
| Claudette Chan | ||||||
| Director and Secretary | ||||||
| Common Stock | 106 | * | ||||
| Class A Preferred | 32 | * | ||||
| Ronald G. Lehman | ||||||
| Director | ||||||
| Common Stock | 4,330 | * | ||||
| Richard E. Turk | ||||||
| Director | ||||||
| Common Stock | 0 | * | ||||
| Jessica Maher | ||||||
| Director | ||||||
| Common Stock | 0 | * | ||||
| All Officers and Directors as a Group (6 persons) | ||||||
| Common Stock | 101,389 | (3) | 1.50 | % | ||
| Class C Stock | 382,447 | 99.98 | % | |||
| Class A Preferred | 2,117 | * | ||||
* Less than one percent
1. Address provided for each beneficial owner owning more than five percent of the voting securities of FONAR.
| Page 89 |
FONAR CORPORATION AND SUBSIDIARIES
ITEM 13. CERTAIN RELATIONSHIPS AND RELATED TRANSACTIONS AND DIRECTOR INDEPENDENCE.
Pursuant to HMCA’s management agreements with its clients, HMCA provides comprehensive non-medical management and administrative services, including billing and collection of accounts, payroll and accounts payable processing, office facilities, supplies and utilities. Under the management agreements, HMCA also provides service for the FONAR Upright® MRI scanners through FONAR. In total, as of September 11, 2023, 22 of our clients had management agreements with HMCA. Six sites in Florida are owned and operated directly by HMCA subsidiaries.
The fees charged under the management agreements are flat fees charged on a monthly basis. These fees ranged from $84,000 to $447,000 per month in fiscal 2023.
Timothy Damadian, the Chairman of the Board, President, Chief Executive Officer and Treasurer of the Company during the 2023 fiscal year owned three of the imaging facilities in Florida managed by HMCA. The facilities were owned by Dr. Raymond Damadian until his death in August 2022. Those facilities paid HMCA flat rate monthly fees ranging from $245,535 to $411,589 per month during fiscal 2023. These fees are renegotiable on an annual basis.
During the fiscal years ended June 30, 2023 and June 30, 2022, the net revenues received by HMCA from the imaging facilities owned by Timothy Damadian and previously by Dr. Damadian were approximately $11.9 million, and $11.6 million, respectively.
Timothy Damadian, the Chairman of the Board, President, Chief Executive Officer and Treasurer of FONAR, was one of the owners of a billing company that performed billing and collection services for No-Fault and Workers’ Compensation claims on behalf of HMCA’s clients. The monthly fee charged to HMCA was $85,000. These services were terminated on January 1, 2021.
On June 1, 2017, the Company also entered into a one year renewable agreement to provide IT services to the billing company for a monthly fee of $23,884. Timothy Damadian is also a Manager of Health Management Company of America. The agreement was renewed on June 1, 2022 and terminated on May 31, 2023, The Company billed them $191,072 in the fiscal year ended June 30, 2023 and $286,608 in the fiscal year ended June 30, 2022.
Timothy Damadian sold his ownership interest in the billing company on May 31, 2023.
Ronald Lehman, a Director of FONAR, holds a .0378% interest in Health Management Company of America’s Class A membership interests.
Claudette J.V. Chan, a Director and the Secretary of FONAR, owns a .0378% interest in Health Management Company of America’s Class A Membership interests.
| Page 90 |
FONAR CORPORATION AND SUBSIDIARIES
ITEM 14. PRINCIPAL ACCOUNTING FEES AND SERVICES.
Audit Fees
The aggregate fees billed by Marcum LLP for the audit of our annual consolidated financial statements for the fiscal year ended June 30, 2023 and the reviews of the financial statements included in our Forms 10-Q for the fiscal year ended June 30, 2023 were $374,000.
The aggregate fees billed by Marcum LLP for the audit of our annual financial statements for the fiscal year ended June 30, 2022 and the reviews of the financial statements included in our Forms 10-Q for the fiscal year ended June 30, 2022 were $379,000.
Audit Related Fees
No fees were billed by Marcum LLP for the fiscal years ended June 30, 2023 or June 30, 2022 for services related to the Audit or review of our financial statements that are not included under the caption “Audit Fees”.
No fees were billed by Marcum LLP for the fiscal years ended June 30, 2023 or June 30, 2022 for designing, operating, supervising or implementing any of our financial information systems or any hardware or software systems for our financial information
Tax Fees
No fees were billed by Marcum LLP for tax compliance, tax advice and tax planning in the fiscal year ended June 30, 2023.
No fees billed by Marcum LLP for tax compliance, tax advice and tax planning in the fiscal year ended June 30, 2022.
All Other Fees
No fees were billed by Marcum LLP for any other services during the fiscal years ended June 30, 2023 and June 30, 2022.
Since January 1, 2003, the audit committee has adopted policies and procedures for pre-approving all non-audit work performed by the auditors. Specifically, the committee must pre-approve the use of the auditors for all such services. The audit committee has pre-approved all non-audit work since that time and in making its determination has considered whether the provision of such services was compatible with the independence of the auditors.
Our audit committee believes that the provision by Marcum LLP of services in addition to audit services in previous years were compatible with maintaining their independence.
| Page 91 |
FONAR CORPORATION AND SUBSIDIARIES
PART IV
ITEM 15. EXHIBITS, FINANCIAL STATEMENT SCHEDULES, AND REPORTS ON FORM 8-K.
a) FINANCIAL STATEMENTS AND SCHEDULES
The following consolidated financial statements are included in Part II, Item 8.
Report of Independent Registered Public Accounting Firm
Consolidated Balance Sheets as at June 30, 2023 and 2022.
Consolidated Statements of Income for the Years Ended June 30, 2023 and 2022.
Consolidated Statements of Stockholders’ Equity for the Years Ended June 30, 2023 and 2022.
Consolidated Statements of Cash Flows for the Years Ended June 30, 2023 and 2022 .
Notes to Consolidated Financial Statements.
Information required by schedules called for under Regulation S-X is either not applicable or is included in the consolidated financial statements or notes to the consolidated financial statements.
b) REPORTS ON FORM 8-K
| c) | EXHIBITS |
3.1 Certificate of Incorporation, as amended, of the Registrant incorporated by reference to Exhibit 3.1 to the Registrant’s registration statement on Form S-1,Commission File No. 33-13365.
| Page 92 |
FONAR CORPORATION AND SUBSIDIARIES
3.5 By-Laws, as amended, of the Registrant incorporated by reference to Exhibit 3.2 to the Registrant’s registration statement on Form S-1, Commission File No. 33-13365.
4.1 Specimen Common Stock Certificate incorporated by reference to Exhibit 4.1 to the Registrant’s registration statement on Form S-1, Commission File No. 33-13365.
4.2 Specimen Class B Common Stock Certificate incorporated by reference to Exhibit 4.2 to the Registrant’s registration statement on Form S-1, Commission File No. 33-13365.
10.1 License Agreement between the Registrant and Raymond V. Damadian incorporated by reference to Exhibit 10 (e) to Form 10-K for the fiscal year ended June 30, 1983, Commission File No. 0-10248.
| Page 93 |
FONAR CORPORATION AND SUBSIDIARIES
21.1 Subsidiaries of the Registrant. See Exhibits.
23.1 Independent Registered Public Accounting Firms Report. See Exhibits.
31.1 Section 302 Certification. See Exhibits.
32.1 Section 906 Certification. See Exhibits.
| Page 94 |
FONAR CORPORATION AND SUBSIDIARIES
SIGNATURES.
Pursuant to the requirements of Section 13 or 15 (d) of the Securities Exchange Act of 1934, the registrant has duly caused this report to be signed on its behalf by the undersigned, thereunto duly authorized.
| FONAR CORPORATION | ||||
| Dated: September 28, 2023 | By: | /s/Timothy Damadian | ||
| Timothy Damadian, | ||||
| Chairman of the Board of Directors | ||||
| Chief Executive Officer | ||||
| President and Treasurer | ||||
| By | /s/Luciano B. Bonanni | |||
| Luciano B. Bonanni | ||||
| Executive Vice President, Chief Operating Officer and Acting Principal Financial Officer |
| Signature | Title | Date | ||
| /s/ Timothy R. Damadian | Chairman of the Board of Directors | September 28, 2023 | ||
| Timothy R. Damadian | Chief Executive Officer | |||
| President and Treasurer | ||||
| /s/Claudette J.V. Chan | Director | September 28, 2023 | ||
| Claudette J.V. Chan | ||||
| /s/Ronald G. Lehman | Director | September 28, 2023 | ||
| Ronald G. Lehman | ||||
| /s/Richard E. Turk | Director | September 28, 2023 | ||
| Richard E. Turk | ||||
| /s/Jessica Maher | Director | September 28, 2023 | ||
| Jessica Maher |
| Page 95 |